- Physics Concept Questions And Answers

Static Electricity Questions
The flow of electrons is referred to as electricity. The imbalance between the electric charges in a body, specifically the imbalance between the negative and the positive charges on a body, is known as static electricity. The electrostatic generator can produce static electricity.
For example, when a comb is rubbed into hair and taken near small pieces of paper, the paper pieces get attracted to the comb.
Read more: Static Electricity
Important Static Electricity Questions with Answers
1. What is electricity?
Answer: The flow of electrons from one section of the circuit to another is known as electricity.
2. Unit of electric power is _____.
Answer: b) Watts
Explanation: The SI unit of power is the watt, equivalent to one joule per second.
3. What is power?
Answer: Power is defined as the rate of doing work; it is the work done in unit time.
4. What is the formula to calculate the power?
- P = W – t
Answer: b) P = W / t
Explanation: Power is given by the formula:
Power = Work / time
5. The electricity built on the surface of a substance is known as ______.
- Current Electricity
- Static electricity
Answer: b) Static electricity
Explanation: When objects are rubbed against each other, static electricity is generated.
6. Define electricity.
Answer: Electricity is defined as the rate of flow of charges in a conductor.
7. How is electric charge classified?
Answer: The electric charge is classified as a positive charge and a negative charge.
8. State true or false: Static electricity is the electric field which is developed due to the stationary charges.
Answer: a) TRUE
Explanation: Electric charges that build up on the surface of materials or substances contribute to static electricity.
9. The fundamental particle of an atom is _____.
- All of the above
Answer: d) All of the above
Explanation: The atom comprises protons, neutrons, and electrons.
10. Static electricity is ______.
- Uncontrolled
Answer: b) Uncontrolled
Explanation: Electrons moving from one body surface to another cannot be controlled.
Practice Questions
- What are the types of electric current?
- Define average power.
- What is meant by the term current electricity?
- Give an example to explain static electricity.
- What is electromotive force?
| PHYSICS Related Links | |
Register with BYJU'S & Download Free PDFs
Register with byju's & watch live videos.

- History & Society
- Science & Tech
- Biographies
- Animals & Nature
- Geography & Travel
- Arts & Culture
- Games & Quizzes
- On This Day
- One Good Fact
- New Articles
- Lifestyles & Social Issues
- Philosophy & Religion
- Politics, Law & Government
- World History
- Health & Medicine
- Browse Biographies
- Birds, Reptiles & Other Vertebrates
- Bugs, Mollusks & Other Invertebrates
- Environment
- Fossils & Geologic Time
- Entertainment & Pop Culture
- Sports & Recreation
- Visual Arts
- Demystified
- Image Galleries
- Infographics
- Top Questions
- Britannica Kids
- Saving Earth
- Space Next 50
- Student Center
- Introduction
How static electricity forms
Examples of static electricity, uses of static electricity.

- What was Michael Faraday’s childhood like?
- Where did Michael Faraday study?
- What did Michael Faraday discover?

static electricity
Our editors will review what you’ve submitted and determine whether to revise the article.
- Online Consortium of Oklahoma - Physical Science - Overview of Static Electricity
- BCcampus Open Publishing - Static Electricity and Charge: Conservation of Charge
- Workforce LibreTexts - Static Electricity
- LiveScience - What is Static Electricity?
- University of Birmingham - Intranet - Static Electricity - shocks and how to avoid them
- Peekskill City School District - Static Electricity
- Table Of Contents

static electricity , form of electricity resulting from the imbalance between positive and negative charges within a material that occurs when electrons (the negatively charged particles in an atom ) move from one material to another. If the electron-receiving material is either isolated or not an electrical conductor , it tends to hold on to the electrons, resulting in a buildup of electric charge . Since this charge is not moving, it is referred to as static electricity. When conditions allow the built-up charge to flow, the surplus of static electricity is discharged, and it becomes current electricity.
When different atoms make contact with one another, electrons can transfer between them. The material shedding electrons also loses negative charge, and it becomes positively charged when there are more protons (positively charged particles) than electrons. Conversely, a material that gains electrons becomes negatively charged. As more electrons move from one material to the other—due to repeated contact between them—additional negative charge will build up in a process that is referred to as the triboelectric effect.
If two objects are rubbed together—especially if the objects are insulators and the surrounding air is dry, such as when a person’s feet move across a carpet—they acquire equal and opposite charges, and an attractive force develops between them. In this example, the atoms in the person’s body strip away electrons from the carpet and leave behind a positive charge on the carpet’s fibres . The electrons become isolated in the atoms of the person’s body. However, when the person gets close to a conducting material, such as when reaching out a hand to touch a doorknob, a path to ground the built-up charge emerges, and electrons jump suddenly to the conducting material. In addition, as the person reaches for the doorknob, some strands of hair may stand on end. Because like charges repel each other, the transferred electrons that build up in the body travel to the extremities—such as individual strands of hair. (This phenomenon can be demonstrated by using a Van de Graaff generator , which uses a moving belt to gather electric charge on a metallic sphere.)
Another example of the effects of static electricity can be observed in a lightning strike, which occurs when a region of a cloud accumulates a surplus of electrical charge. Small hail particles form in a cloud when moisture in the air freezes, and these particles transfer charge as they grow, move within the cloud, and collide with one another. As additional charged hail particles form, a charge separation occurs, with air acting as an insulator between the charged particles in the cloud as well as between the cloud and the ground. When the surplus of charge is great enough, it overcomes the insulating ability of the air, and electricity is discharged between clouds or between the ground and the cloud itself.
Some of the best-known uses of static electricity occur in air filters and dust-removal devices, which take advantage of the charge differences between materials to remove airborne particles. As electrostatically charged air particles pass through the filter system, the layers of the filter, which have an opposite charge, pick them up and trap them. The buildup of static charge is not always beneficial , however. It can cause damage to important electrical components in computer chips and other components in circuits. Additionally, the friction that occurs while pumping liquids through hoses or pipelines can cause a static charge to accumulate, which can be hazardous if these liquids or the gases they produce are flammable. When this static charge comes into contact with a grounded object, it can create a spark that could ignite these materials.
- TPC and eLearning
- What's NEW at TPC?
- Read Watch Interact
- Practice Review Test
- Teacher-Tools
- Request a Demo
- Get A Quote
- Subscription Selection
- Seat Calculator
- Ad Free Account
- Edit Profile Settings
- Metric Conversions Questions
- Metric System Questions
- Metric Estimation Questions
- Significant Digits Questions
- Proportional Reasoning
- Acceleration
- Distance-Displacement
- Dots and Graphs
- Graph That Motion
- Match That Graph
- Name That Motion
- Motion Diagrams
- Pos'n Time Graphs Numerical
- Pos'n Time Graphs Conceptual
- Up And Down - Questions
- Balanced vs. Unbalanced Forces
- Change of State
- Force and Motion
- Mass and Weight
- Match That Free-Body Diagram
- Net Force (and Acceleration) Ranking Tasks
- Newton's Second Law
- Normal Force Card Sort
- Recognizing Forces
- Air Resistance and Skydiving
- Solve It! with Newton's Second Law
- Which One Doesn't Belong?
- Component Addition Questions
- Head-to-Tail Vector Addition
- Projectile Mathematics
- Trajectory - Angle Launched Projectiles
- Trajectory - Horizontally Launched Projectiles
- Vector Addition
- Vector Direction
- Which One Doesn't Belong? Projectile Motion
- Forces in 2-Dimensions
- Being Impulsive About Momentum
- Explosions - Law Breakers
- Hit and Stick Collisions - Law Breakers
- Case Studies: Impulse and Force
- Impulse-Momentum Change Table
- Keeping Track of Momentum - Hit and Stick
- Keeping Track of Momentum - Hit and Bounce
- What's Up (and Down) with KE and PE?
- Energy Conservation Questions
- Energy Dissipation Questions
- Energy Ranking Tasks
- LOL Charts (a.k.a., Energy Bar Charts)
- Match That Bar Chart
- Words and Charts Questions
- Name That Energy
- Stepping Up with PE and KE Questions
- Case Studies - Circular Motion
- Circular Logic
- Forces and Free-Body Diagrams in Circular Motion
- Gravitational Field Strength
- Universal Gravitation
- Angular Position and Displacement
- Linear and Angular Velocity
- Angular Acceleration
- Rotational Inertia
- Balanced vs. Unbalanced Torques
- Getting a Handle on Torque
- Torque-ing About Rotation
- Properties of Matter
- Fluid Pressure
- Buoyant Force
- Sinking, Floating, and Hanging
- Pascal's Principle
- Flow Velocity
- Bernoulli's Principle
- Balloon Interactions
- Charge and Charging
- Charge Interactions
- Charging by Induction
- Conductors and Insulators
- Coulombs Law
- Electric Field
- Electric Field Intensity
- Polarization
- Case Studies: Electric Power
- Know Your Potential
- Light Bulb Anatomy
- I = ∆V/R Equations as a Guide to Thinking
- Parallel Circuits - ∆V = I•R Calculations
- Resistance Ranking Tasks
- Series Circuits - ∆V = I•R Calculations
- Series vs. Parallel Circuits
- Equivalent Resistance
- Period and Frequency of a Pendulum
- Pendulum Motion: Velocity and Force
- Energy of a Pendulum
- Period and Frequency of a Mass on a Spring
- Horizontal Springs: Velocity and Force
- Vertical Springs: Velocity and Force
- Energy of a Mass on a Spring
- Decibel Scale
- Frequency and Period
- Closed-End Air Columns
- Name That Harmonic: Strings
- Rocking the Boat
- Wave Basics
- Matching Pairs: Wave Characteristics
- Wave Interference
- Waves - Case Studies
- Color Addition and Subtraction
- Color Filters
- If This, Then That: Color Subtraction
- Light Intensity
- Color Pigments
- Converging Lenses
- Curved Mirror Images
- Law of Reflection
- Refraction and Lenses
- Total Internal Reflection
- Who Can See Who?
- Lab Equipment
- Lab Procedures
- Formulas and Atom Counting
- Atomic Models
- Bond Polarity
- Entropy Questions
- Cell Voltage Questions
- Heat of Formation Questions
- Reduction Potential Questions
- Oxidation States Questions
- Measuring the Quantity of Heat
- Hess's Law
- Oxidation-Reduction Questions
- Galvanic Cells Questions
- Thermal Stoichiometry
- Molecular Polarity
- Quantum Mechanics
- Balancing Chemical Equations
- Bronsted-Lowry Model of Acids and Bases
- Classification of Matter
- Collision Model of Reaction Rates
- Density Ranking Tasks
- Dissociation Reactions
- Complete Electron Configurations
- Elemental Measures
- Enthalpy Change Questions
- Equilibrium Concept
- Equilibrium Constant Expression
- Equilibrium Calculations - Questions
- Equilibrium ICE Table
- Intermolecular Forces Questions
- Ionic Bonding
- Lewis Electron Dot Structures
- Limiting Reactants
- Line Spectra Questions
- Mass Stoichiometry
- Measurement and Numbers
- Metals, Nonmetals, and Metalloids
- Metric Estimations
- Metric System
- Molarity Ranking Tasks
- Mole Conversions
- Name That Element
- Names to Formulas
- Names to Formulas 2
- Nuclear Decay
- Particles, Words, and Formulas
- Periodic Trends
- Precipitation Reactions and Net Ionic Equations
- Pressure Concepts
- Pressure-Temperature Gas Law
- Pressure-Volume Gas Law
- Chemical Reaction Types
- Significant Digits and Measurement
- States Of Matter Exercise
- Stoichiometry Law Breakers
- Stoichiometry - Math Relationships
- Subatomic Particles
- Spontaneity and Driving Forces
- Gibbs Free Energy
- Volume-Temperature Gas Law
- Acid-Base Properties
- Energy and Chemical Reactions
- Chemical and Physical Properties
- Valence Shell Electron Pair Repulsion Theory
- Writing Balanced Chemical Equations
- Mission CG1
- Mission CG10
- Mission CG2
- Mission CG3
- Mission CG4
- Mission CG5
- Mission CG6
- Mission CG7
- Mission CG8
- Mission CG9
- Mission EC1
- Mission EC10
- Mission EC11
- Mission EC12
- Mission EC2
- Mission EC3
- Mission EC4
- Mission EC5
- Mission EC6
- Mission EC7
- Mission EC8
- Mission EC9
- Mission RL1
- Mission RL2
- Mission RL3
- Mission RL4
- Mission RL5
- Mission RL6
- Mission KG7
- Mission RL8
- Mission KG9
- Mission RL10
- Mission RL11
- Mission RM1
- Mission RM2
- Mission RM3
- Mission RM4
- Mission RM5
- Mission RM6
- Mission RM8
- Mission RM10
- Mission LC1
- Mission RM11
- Mission LC2
- Mission LC3
- Mission LC4
- Mission LC5
- Mission LC6
- Mission LC8
- Mission SM1
- Mission SM2
- Mission SM3
- Mission SM4
- Mission SM5
- Mission SM6
- Mission SM8
- Mission SM10
- Mission KG10
- Mission SM11
- Mission KG2
- Mission KG3
- Mission KG4
- Mission KG5
- Mission KG6
- Mission KG8
- Mission KG11
- Mission F2D1
- Mission F2D2
- Mission F2D3
- Mission F2D4
- Mission F2D5
- Mission F2D6
- Mission KC1
- Mission KC2
- Mission KC3
- Mission KC4
- Mission KC5
- Mission KC6
- Mission KC7
- Mission KC8
- Mission AAA
- Mission SM9
- Mission LC7
- Mission LC9
- Mission NL1
- Mission NL2
- Mission NL3
- Mission NL4
- Mission NL5
- Mission NL6
- Mission NL7
- Mission NL8
- Mission NL9
- Mission NL10
- Mission NL11
- Mission NL12
- Mission MC1
- Mission MC10
- Mission MC2
- Mission MC3
- Mission MC4
- Mission MC5
- Mission MC6
- Mission MC7
- Mission MC8
- Mission MC9
- Mission RM7
- Mission RM9
- Mission RL7
- Mission RL9
- Mission SM7
- Mission SE1
- Mission SE10
- Mission SE11
- Mission SE12
- Mission SE2
- Mission SE3
- Mission SE4
- Mission SE5
- Mission SE6
- Mission SE7
- Mission SE8
- Mission SE9
- Mission VP1
- Mission VP10
- Mission VP2
- Mission VP3
- Mission VP4
- Mission VP5
- Mission VP6
- Mission VP7
- Mission VP8
- Mission VP9
- Mission WM1
- Mission WM2
- Mission WM3
- Mission WM4
- Mission WM5
- Mission WM6
- Mission WM7
- Mission WM8
- Mission WE1
- Mission WE10
- Mission WE2
- Mission WE3
- Mission WE4
- Mission WE5
- Mission WE6
- Mission WE7
- Mission WE8
- Mission WE9
- Vector Walk Interactive
- Name That Motion Interactive
- Kinematic Graphing 1 Concept Checker
- Kinematic Graphing 2 Concept Checker
- Graph That Motion Interactive
- Two Stage Rocket Interactive
- Rocket Sled Concept Checker
- Force Concept Checker
- Free-Body Diagrams Concept Checker
- Free-Body Diagrams The Sequel Concept Checker
- Skydiving Concept Checker
- Elevator Ride Concept Checker
- Vector Addition Concept Checker
- Vector Walk in Two Dimensions Interactive
- Name That Vector Interactive
- River Boat Simulator Concept Checker
- Projectile Simulator 2 Concept Checker
- Projectile Simulator 3 Concept Checker
- Hit the Target Interactive
- Turd the Target 1 Interactive
- Turd the Target 2 Interactive
- Balance It Interactive
- Go For The Gold Interactive
- Egg Drop Concept Checker
- Fish Catch Concept Checker
- Exploding Carts Concept Checker
- Collision Carts - Inelastic Collisions Concept Checker
- Its All Uphill Concept Checker
- Stopping Distance Concept Checker
- Chart That Motion Interactive
- Roller Coaster Model Concept Checker
- Uniform Circular Motion Concept Checker
- Horizontal Circle Simulation Concept Checker
- Vertical Circle Simulation Concept Checker
- Race Track Concept Checker
- Gravitational Fields Concept Checker
- Orbital Motion Concept Checker
- Angular Acceleration Concept Checker
- Balance Beam Concept Checker
- Torque Balancer Concept Checker
- Aluminum Can Polarization Concept Checker
- Charging Concept Checker
- Name That Charge Simulation
- Coulomb's Law Concept Checker
- Electric Field Lines Concept Checker
- Put the Charge in the Goal Concept Checker
- Circuit Builder Concept Checker (Series Circuits)
- Circuit Builder Concept Checker (Parallel Circuits)
- Circuit Builder Concept Checker (∆V-I-R)
- Circuit Builder Concept Checker (Voltage Drop)
- Equivalent Resistance Interactive
- Pendulum Motion Simulation Concept Checker
- Mass on a Spring Simulation Concept Checker
- Particle Wave Simulation Concept Checker
- Boundary Behavior Simulation Concept Checker
- Slinky Wave Simulator Concept Checker
- Simple Wave Simulator Concept Checker
- Wave Addition Simulation Concept Checker
- Standing Wave Maker Simulation Concept Checker
- Color Addition Concept Checker
- Painting With CMY Concept Checker
- Stage Lighting Concept Checker
- Filtering Away Concept Checker
- InterferencePatterns Concept Checker
- Young's Experiment Interactive
- Plane Mirror Images Interactive
- Who Can See Who Concept Checker
- Optics Bench (Mirrors) Concept Checker
- Name That Image (Mirrors) Interactive
- Refraction Concept Checker
- Total Internal Reflection Concept Checker
- Optics Bench (Lenses) Concept Checker
- Kinematics Preview
- Velocity Time Graphs Preview
- Moving Cart on an Inclined Plane Preview
- Stopping Distance Preview
- Cart, Bricks, and Bands Preview
- Fan Cart Study Preview
- Friction Preview
- Coffee Filter Lab Preview
- Friction, Speed, and Stopping Distance Preview
- Up and Down Preview
- Projectile Range Preview
- Ballistics Preview
- Juggling Preview
- Marshmallow Launcher Preview
- Air Bag Safety Preview
- Colliding Carts Preview
- Collisions Preview
- Engineering Safer Helmets Preview
- Push the Plow Preview
- Its All Uphill Preview
- Energy on an Incline Preview
- Modeling Roller Coasters Preview
- Hot Wheels Stopping Distance Preview
- Ball Bat Collision Preview
- Energy in Fields Preview
- Weightlessness Training Preview
- Roller Coaster Loops Preview
- Universal Gravitation Preview
- Keplers Laws Preview
- Kepler's Third Law Preview
- Charge Interactions Preview
- Sticky Tape Experiments Preview
- Wire Gauge Preview
- Voltage, Current, and Resistance Preview
- Light Bulb Resistance Preview
- Series and Parallel Circuits Preview
- Thermal Equilibrium Preview
- Linear Expansion Preview
- Heating Curves Preview
- Electricity and Magnetism - Part 1 Preview
- Electricity and Magnetism - Part 2 Preview
- Vibrating Mass on a Spring Preview
- Period of a Pendulum Preview
- Wave Speed Preview
- Slinky-Experiments Preview
- Standing Waves in a Rope Preview
- Sound as a Pressure Wave Preview
- DeciBel Scale Preview
- DeciBels, Phons, and Sones Preview
- Sound of Music Preview
- Shedding Light on Light Bulbs Preview
- Models of Light Preview
- Electromagnetic Radiation Preview
- Electromagnetic Spectrum Preview
- EM Wave Communication Preview
- Digitized Data Preview
- Light Intensity Preview
- Concave Mirrors Preview
- Object Image Relations Preview
- Snells Law Preview
- Reflection vs. Transmission Preview
- Magnification Lab Preview
- Reactivity Preview
- Ions and the Periodic Table Preview
- Periodic Trends Preview
- Chemical Reactions Preview
- Intermolecular Forces Preview
- Melting Points and Boiling Points Preview
- Bond Energy and Reactions Preview
- Reaction Rates Preview
- Ammonia Factory Preview
- Stoichiometry Preview
- Nuclear Chemistry Preview
- Gaining Teacher Access
- Task Tracker Directions
- Conceptual Physics Course
- On-Level Physics Course
- Honors Physics Course
- Chemistry Concept Builders
- All Chemistry Resources
- Users Voice
- Tasks and Classes
- Webinars and Trainings
- Subscription
- Subscription Locator
- 1-D Kinematics
- Newton's Laws
- Vectors - Motion and Forces in Two Dimensions
- Momentum and Its Conservation
- Work and Energy
- Circular Motion and Satellite Motion
- Thermal Physics
- Static Electricity
- Electric Circuits
- Vibrations and Waves
- Sound Waves and Music
- Light and Color
- Reflection and Mirrors
- Measurement and Calculations
- About the Physics Interactives
- Task Tracker
- Usage Policy
- Newtons Laws
- Vectors and Projectiles
- Forces in 2D
- Momentum and Collisions
- Circular and Satellite Motion
- Balance and Rotation
- Electromagnetism
- Waves and Sound
- Atomic Physics
- Forces in Two Dimensions
- Work, Energy, and Power
- Circular Motion and Gravitation
- Sound Waves
- 1-Dimensional Kinematics
- Circular, Satellite, and Rotational Motion
- Einstein's Theory of Special Relativity
- Waves, Sound and Light
- QuickTime Movies
- About the Concept Builders
- Pricing For Schools
- Directions for Version 2
- Measurement and Units
- Relationships and Graphs
- Rotation and Balance
- Vibrational Motion
- Reflection and Refraction
- Teacher Accounts
- Kinematic Concepts
- Kinematic Graphing
- Wave Motion
- Sound and Music
- About CalcPad
- 1D Kinematics
- Vectors and Forces in 2D
- Simple Harmonic Motion
- Rotational Kinematics
- Rotation and Torque
- Rotational Dynamics
- Electric Fields, Potential, and Capacitance
- Transient RC Circuits
- Light Waves
- Units and Measurement
- Stoichiometry
- Molarity and Solutions
- Thermal Chemistry
- Acids and Bases
- Kinetics and Equilibrium
- Solution Equilibria
- Oxidation-Reduction
- Nuclear Chemistry
- Newton's Laws of Motion
- Work and Energy Packet
Static Electricity Review
- NGSS Alignments
- 1D-Kinematics
- Projectiles
- Circular Motion
- Magnetism and Electromagnetism
- Graphing Practice
- About the ACT
- ACT Preparation
- For Teachers
- Other Resources
- Solutions Guide
- Solutions Guide Digital Download
- Motion in One Dimension
- Work, Energy and Power
- Chemistry of Matter
- Names and Formulas
- Algebra Based On-Level Physics
- Honors Physics
- Conceptual Physics
- Other Tools
- Frequently Asked Questions
- Purchasing the Download
- Purchasing the Digital Download
- About the NGSS Corner
- NGSS Search
- Force and Motion DCIs - High School
- Energy DCIs - High School
- Wave Applications DCIs - High School
- Force and Motion PEs - High School
- Energy PEs - High School
- Wave Applications PEs - High School
- Crosscutting Concepts
- The Practices
- Physics Topics
- NGSS Corner: Activity List
- NGSS Corner: Infographics
- About the Toolkits
- Position-Velocity-Acceleration
- Position-Time Graphs
- Velocity-Time Graphs
- Newton's First Law
- Newton's Second Law
- Newton's Third Law
- Terminal Velocity
- Projectile Motion
- Forces in 2 Dimensions
- Impulse and Momentum Change
- Momentum Conservation
- Work-Energy Fundamentals
- Work-Energy Relationship
- Roller Coaster Physics
- Satellite Motion
- Electric Fields
- Circuit Concepts
- Series Circuits
- Parallel Circuits
- Describing-Waves
- Wave Behavior Toolkit
- Standing Wave Patterns
- Resonating Air Columns
- Wave Model of Light
- Plane Mirrors
- Curved Mirrors
- Teacher Guide
- Using Lab Notebooks
- Current Electricity
- Light Waves and Color
- Reflection and Ray Model of Light
- Refraction and Ray Model of Light
- Teacher Resources
- Subscriptions

- Newton's Laws
- Einstein's Theory of Special Relativity
- About Concept Checkers
- School Pricing
- Newton's Laws of Motion
- Newton's First Law
- Newton's Third Law
Navigate to:
Review Session Home - Topic Listing Static Electricity - Home || Printable Version || Questions with Links Answers to Questions: All || #1-16 || #17-32 || #33-43
[ #1 | #2 | #3 | #4 | #5 | #6 | #7 | #8 | #9 | #10 | #11 | #12 | #13 | #14 | #15 | #16 | #17 | #18 | #19 | #20 | #21 | #22 | #23 | #24 | #25 | #26 | #27 | #28 | #29 | #30 | #31 | #32 | #33 | #34 | #35 | #36 | #37 | #38 | #39 | #40 | #41 | #42 | #43 ]
Part A: Multiple Choice
1. Which of the following are true of static charges? Choose all that apply.
- Like charges repel.
- Like charges attract.
- Opposite charges repel.
- Opposite charges attract.
- A positively charged object has lost electrons.
- A positively charged object has gained protons.
- A negatively charged object has lost protons.
- A negatively charged object has gained electrons.
| || || [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
2. Identify the following objects as being either
a. positive b. negative c. neutral If there is no conclusive evidence, then select all that could be true. Description of Object a, b, or c? i. An object possesses more protons than electrons. ii. An object possesses more neutrons than electrons. iii. A formerly neutral object that just lost some electrons. iv. A formerly neutral object that just gained some electrons. v. An object which attracts a negatively-charged balloon. vi. An object which attracts neutral paper bits and attracts a negatively-charged balloon. vii. An object which attracts neutral attracts paper bits and repels a negatively-charged balloon. viii. An object which attracts a negatively-charged balloon and attracts a positively-charged balloon. ix. An object which attracts a charged balloon (balloon A) which is attracted to a negatively-charged balloon (balloon B). x. An object which attracts a balloon (balloon C) which is repelled by a negatively-charged balloon (balloon D). xi. An object which repels a balloon (balloon E) which is repelled by a positively-charged balloon (balloon F). xii. An object around which the electric field vector is directed inwards. xiii. An object around which the electric field vector is directed outwards.
| || || | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
3. Consider the following statements given below and determine if the charge of Object A is:
| a. positive | b. negative | c. neutral |
Description of Charging Method a, b, or c? i. Object A is charged by friction using animal fur. Animal fur has a greater electron affinity than object A. ii. Object A is charged by contact using a negatively charged object. iii. Object A is charged by induction using a positively charged object. iv. Object A is used to charge an aluminum plate by the method of induction. The aluminum plate acquires a positive charge. v. Object A is used to charge an aluminum plate by the method of induction. The aluminum plate acquires a negative charge. vi. A rubber rod has a greater electron affinity than animal fur. The rubber rod is charged by friction with animal fur. The rubber rod is then used to charge Object A by the method of contact. vii. A rubber rod has a greater electron affinity than animal fur. The rubber rod is charged by friction with animal fur. The rubber rod is then used to charge Object A by the method of induction. viii. A rubber rod has a greater electron affinity than animal fur. The rubber rod is charged by friction with animal fur. The rubber rod is then used to charge an aluminum pop can by the process of induction. The pop can is then contacted to object A.
4. A neutral plastic strip is rubbed with cotton and acquires a positive charge. Which of the following statements are true of the positively-charged strip?
- It lost some electrons to the cotton during the charging process.
- It lost all of its electrons to the cotton during the charging process.
- It has the opposite charge as the cotton.
- It would now be repelled by the piece of cotton which was used to charge it.
- It gained protons during the rubbing process.
- As a material, plastic has a greater affinity for electrons than cotton.
- It could exert either a repulsive or attractive influence upon neutral paper bits.
- It has an excess of protons compared to the number of electrons.
- It could be used to charge an electroscope negatively by the process of induction.
- It lost negative electrons and gained positive electrons during the charging process.
- It lost neutrons during the charging process (or at the very least, its neutrons became ineffective).
5. A positively-charged glass rod is touched to the plate of a neutral electroscope. Upon contact, the electroscope becomes charged and the needle deflects. Which of the following statements are true of the charged electroscope?
- The electroscope is now charged positively.
- The electroscope and the glass rod now have the same type of charge.
- The electroscope was charged by the method of induction.
- The electroscope gained protons during the charging process.
- The electroscope gained electrons during the charging process.
- The electroscope lost all of its electrons during the charging process.
- During the charging process, some electrons left the electroscope and entered the glass rod.
- The number of electrons present in the electroscope is less than the number of protons.
- The electroscope needle will deflect even more if the glass rod is brought near it again.
- The electroscope needle would slowly approach the neutral position if a negatively-charged balloon is brought near.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
6. A negatively-charged balloon is brought near to (without touching) a neutral electroscope. With the negatively-charged balloon held near, the electroscope is momentarily touched by a hand ( ground ). The balloon is then removed and the needle deflects, thus showing a charge. Which of the following statements are true of the charged electroscope?
- The charge on the electroscope is the same type of the charge on the balloon.
- The electroscope has more protons than electrons.
- The electroscope lost some electrons during the charging process.
- During the charging process, protons moved from the electroscope to the balloon.
- During the charging process, electrons moved from the electroscope to the balloon.
- During the charging process, electrons moved from the electroscope to the hand (ground).
- During the charging process, electrons moved from the hand (ground) to the electroscope.
7. The outer shell electrons in metals are not tightly bound to the nuclei of their atoms. They are free to roam throughout the material, moving from atom to atom. These materials are good ____.
- for nothing
8. The charge on the metal sphere will end up ___.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
9. The sphere acquires this charge because ____.
- electrons move from the rubber rod to the sphere
- electrons move from the sphere to the rubber rod
- protons move from the rubber rod to the sphere
- protons move from the sphere to the rubber rod
- the rubber rod creates a charge on the sphere
10. If a positively charged plate is brought near the top of a positively-charged electroscope, then the deflected needle will ____.
- not move at all
- be deflected more
- be deflected less
| || | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
11. If an electroscope, charged up with excess negative charge, is touched and grounded, then it will end up with ___ charge.
12. A negatively charged balloon is brought near a metal can that rests on a wood table. The side of the can opposite the balloon is momentarily touched. The can is then _______.
- positively charged
- negatively charged
13. Charge carriers in a metal are electrons rather than protons. This is due to the fact that electrons are __________.
- loosely bound
- far from a nucleus
- all of the above
- none of the above
14. Two like charges ________.
- attract each other
- repel each other
- neutralize each other
- have no effect on each other
- must be neutrons
15. If you comb your hair and the comb becomes positively charged, then your hair becomes _______.
- uncharged
16. Which of the following charging methods can result in charging an object with a negative charge? Choose all that apply.
| a. charging by friction | b. charging by contact | c. charging by induction |
17. Which of the following charging methods work without ever touching the object to be charged to the object used to charge it? Choose all that apply.
| || || [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
18. Which of the following charging methods result in charging an object opposite to the charge on the object used to charge it? Choose all that apply.
| || || [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
19. Which of the following statements are true statements about electric force? Choose all that apply.
- Electric force is a contact force.
- Electric forces can only act between charged objects - either like-charged or oppositely-charged.
- Electric forces between two charged objects increases with increasing separation distance.
- Electric forces between two charged objects increases with increasing quantity of charge on the objects.
- If object A attracts object B with an electric force, then the attractive force must be mutual - i.e., object B also attracts object A with the same force.
- A doubling of the quantity of charge on one of the objects results in a doubling of the electric force.
- A tripling of the quantity of charge on both of the objects results in an increase in the electric force by a factor of 6.
- A doubling of the separation distance between two point charges results in a quadrupling of the electric force.
- A tripling of the separation distance between two point charges results in an electric force which is one-sixth of the original value.
20. Which of the following statements are true statements about electric field? Choose all that apply.
- The electric field strength created by object A is dependent upon the separation distance from object A.
- The electric field strength created by object A is dependent upon the charge on object A.
- The electric field strength created by object A is dependent upon the charge of the test object used to measure the strength of the field.
- The electric field strength about charged Object A is the force per charge experienced by a test charge placed at some location about Object A.
- As the distance from Object A is doubled, the electric field strength created by object A increases by a factor of 4.
- As the charge of Object A is doubled, the electric field strength created by object A increases by a factor of 2.
- As the charge of Object A is doubled and the distance from object A is doubled, the electric field strength created by object A decreases by a factor of 2.
- Object B is used to test the electric field strength about Object A; as the charge of Object B is doubled, the force which it experiences is doubled but the electric field strength remains the same.
- Object B is used to test the electric field strength about Object A; as the separation distance between Object A and Object B is doubled, the force which it experiences decreases by a factor of 4 but the electric field strength remains the same.
- The electric field strength inside of a closed conducting object (e.g., inside the sphere of the Van de Graaff generator) is zero.
- For irregularly shaped objects, the electric field strength is greatest around points of highest curvature.
- The electric field is a vector which points in the direction which a positive test charge would be accelerated.
- Electric fields are directed inwards in regions around negatively-charged objects and outwards in regions around positively-charged objects.
- Units on electric field are Newtons/Coulomb (N/C).
| || || [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
21. Which of the following statements are true statements about lightning rods? Choose all that apply.
- Lightning rods are placed on homes to reduce the risk of lightning damage to a home.
- Any metallic object which is placed on the roof of a home and grounded by an appropriate conducting pathway can serve as a lightning rod.
- Most lightning rods are pointed as a decorative feature.
- To be totally effective, a lightning rod must stretch high into the sky and draw charge from the lowest clouds by the method of contact.
- Lightning rods are capable of reducing the excess charge buildup in clouds, a characteristic of dangerous thunderstorms.
- Charge is incapable of passing from clouds to a lightning rods since the air between the clouds and the lightning rod has an insulating effect.
- The electric field strength about the points of lightning rods are very high.
| || [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
22. Charged balloons are used to induce a charge upon neutral metal pop cans. Identify the type of excess charge which would be present on cans G, H, I, J, K, L, M, N and P as shown in the diagrams below.
a. positive b. negative c. neutral
| I. | ||
| II. | ||
| II. | M: ____ | |
| IV. | ||
| V. |
| [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
23. In diagram IV of Question #22 , the pop can acquires the charge that it does because ___.
- electrons move from the balloon to the pop can
- electrons move from the pop can to the balloon
- protons move from the balloon to the pop can
- protons move from the pop can to the balloon
- electrons move from the pop can to the hand ( ground )
- electrons move from the hand ( ground ) to the pop can
- protons move from the pop can to the hand ( ground )
- protons move from the hand ( ground ) to the pop can
24. A negatively charged balloon will be attracted to a neutral wooden cabinet due to polarization. Which one of the following diagrams best depict why this occurs?
For Questions #25 - #27 , identify the type of charge on objects A-D based on the electric field lines shown for each configuration of charges.
| 25. | 26. |
| 27. | |
| [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
Part B: Diagramming and Analysis
28. A neutral conducting sphere is charged by induction using a positively-charged balloon. What will be the charge of the following sphere in step e if steps a - d are followed? _________
Explain how the object acquires this charge. Show the type and location of excess charge on the conducting sphere in each step of the diagram (where appropriate).
| [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
a. Draw the location and type of excess charges on the "polarized" neutral electroscope.
b. Explain how the balloon has induced the temporary charge upon the electroscope (i.e., describe the direction of electron movement).
| |
[ #1 | #2 | #3 | #4 | #5 | #6 | #7 | #8 | #9 | #10 | #11 | #12 | #13 | #14 | #15 | #16 | #17 | #18 | #19 | #20 | #21 | #22 | #23 | #24 | #25 | #26 | #27 | #28 | #29 | #30 | #31 | #32 | #33 | #34 | #35 | #36 | #37 | #38 | #39 | #40 | #41 | #42 | #43 ]
30. Construct electric field lines around the following configuration of charges. Include at least six lines per charge.
| a. | b. |
| c. | d. |
31. Use an unbroken arrow to show the direction of electron movement in the following situation. The arrow should extend from the source of the electrons to the final destination of the electrons.
| a. A positively charged balloon is touched to a neutral conducting sphere. | b. A glass rod is rubbed with a piece of wool. The wool has the greater electron affinity. |
| c. A positively-charged balloon is held near a neutral conducting sphere. The sphere is then touched on the opposite side. | d. A negatively-charged rod is held near a neutral conducting sphere. The sphere is touched on the opposite side. |
| || || [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ]
|
32. A negatively-charged object creates an electric field which can be measured at various locations in the region about it. An electric field vector is drawn for point X. Use your understanding of the electric field - distance relationship to draw E vectors for points A - E. (The length of the arrow should be indicative of the relative strength of the E vector.)
Part C: Short Computations
33. The charge of one electron is -1.6 X 10 -19 Coulombs. If a neutral object loses 1.5x10 6 electrons, then what will be its charge?
34. Express your understanding of Coulomb's law by filling in the following table.
| (C or µC) | (C or µC) | (N) | ||
| a. | C | C | ||
| b. | C | C | ||
| c. | ||||
| d. | ||||
| e. | ) | N |
35. Charged object A ( Q A ) creates an electric field ( E ). A positively-charged test charge ( q B ) is used to measure the electric force ( F ) at various distances ( d ) from object A. Express your understanding of electric field strength and electric force by filling in the following table. (Note that the units on charge are fictitious units. Since the value of k is not known in these units, you will have to use your qualitative understanding to answer this question. That is, you will have to think about relationships - the effect that doubling or quadrupling or halving ... the d or the Q has upon the force and the field strength.)
| (bP) | (bP) | ||||
| a. | |||||
| b. | |||||
| c. | |||||
| d. | |||||
| e. | |||||
| f. | |||||
| g. |
36. The following questions check your qualitative understanding of the variables effecting electric field and electric force. Use your understanding to fill in the blanks.
- At a separation distance of 0.500-m, two like-charged balloons experience a repulsive force of 0.320 N. If the distance is doubled, then the repulsive force would be ______ N.
- At a separation distance of 0.500-m, two like-charged balloons experience a repulsive force of 0.320 N. If the distance is halved, then the repulsive force would be ______ N.
- At a separation distance of 0.500-m, two like-charged balloons experience a repulsive force of 0.320 N. If the distance is is decreased by a factor of 3 and the charge on one of the balloons is doubled, then the repulsive force would be ______ N.
- At a separation distance of 0.500-m, two like-charged balloons experience a repulsive force of 0.320 N. If the distance is is increased by a factor of 3 and the charge on both of the balloons is doubled, then the repulsive force would be ______ N.
- At a location of 0.200 m from object A, the electric field strength is 0.0500 N/C. If the electric field strength were measured at a location of 0.40 m from object A, then the field strength would be _______ N/C.
- At a location of 0.200 m from object A, the electric field strength is 0.0500 N/C. If the electric field strength were measured at a location of 1.000 m from object A, then the field strength would be _______ N/C
- At a location of 0.200 m from object A, the electric field strength is 0.0500 N/C. If the electric field strength were measured using a test charge with twice the charge at a location of 0.200 m from object A, then the field strength would be _______ N/C
- At a location of 0.200 m from object A, the electric field strength is 0.0500 N/C. If the electric field strength were measured using a test charge with twice the charge at a location of 0.100 m from object A, then the field strength would be _______ N/C.
Part D: Problem-Solving
37. Two objects are located along the y-axis. Object A has a charge of +25 µC and is located at the origin. Object B has a charge of -16 µC and is located a distance of 62 cm above object A. Determine the y-coordinate location where the electric field is zero.
38. Three objects are located along the x-axis. Object A with a charge of +5.6 µC is located at the origin. Object B has a charge of -4.2 µC and is located at the -1.2 m location. Object C has a charge of +7.7 µC and is located at the +2.4 m location. Determine the magnitude and direction of the net electric force acting upon object A.
39. Object A has a x-y coordinate position of (+5.0, 0.0). Object B has a x-y coordinate location of (0.0, +4.0). If object A has a charge of -5.8 µC and object B has a charge of +8.9 µC, then what is the resultant electric field strength at the origin.
| [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
40. For the situation described in Question #39 , what would be the magnitude of the net electric force upon a +1.3 µC charge if placed at the origin.
41. Alpha particles (i.e., Helium nucleus) have a molar mass of 4.002 g/mol and consist of two protons and two neutrons.
- Determine the charge of one alpha particle in units of Coulombs and the mass of one alpha particle in units of kg.
- Suppose that Ernie Rufferthord (not to be confused with the scientist of gold foil fame) wishes to suspend an alpha particle in midair by attracting it to a bundle of electrons held a distance of 1.00-m above the alpha particle. How many electrons would Ernie need in his bundle to accomplish such an amazing feat?
42. A 1.19-gram charged balloon hangs from a 1.99-m string which is attached to the ceiling. A Van de Graaff generator is located directly below the location where the string attaches to the ceiling and is at the same height as the balloon. The string is deflected at an angle of 32.0 degrees from the vertical due to the presence of the electric field. Determine the charge on the Van de Graaff generator if the charge on the balloon is 2.27x10 -12 C.
43. Ignoring trace elements, a typical elemental composition (by mass) of the human body is as follows:
- Use these percentages, the molar mass values, Avogadro's number, and the atomic numbers to determine the total number of electrons (and protons) in a 73-kg human body (160 pounds).
- If these electrons and protons were placed 100 m apart (the distance of approximately one football field), then what would be the force of electrical attraction between them.
| [ | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ] |
Review Session Home - Topic Listing Static Electricity - Home || Printable Version || Questions with Links Answers to Questions: All || #1-16 || #17-32 || #33-43
You Might Also Like ...
- The Calculator Pad The Calculator Pad includes physics word problems organized by topic. Each problem is accompanied by a pop-up answer and an audio file that explains the details of how to approach and solve the problem. It's a perfect resource for those wishing to improve their problem-solving skills. Visit: The Calculator Pad Home | Calculator Pad - Static Electricity
- Minds On Physics the App Series Minds On Physics the App ("MOP the App") is a series of interactive questioning modules for the student that is serious about improving their conceptual understanding of physics. Each module of the series covers a different topic and is further broken down into sub-topics. A "MOP experience" will provide a learner with challenging questions, feedback, and question-specific help in the context of a game-like environment. It is available for phones, tablets, Chromebooks, and Macintosh computers. It's a perfect resource for those wishing to refine their conceptual reasoning abilities. Part 4 of the series includes topics in Static Electricity. Visit: MOP the App Home || MOP the App - Part 4

Want to create or adapt books like this? Learn more about how Pressbooks supports open publishing practices.
Electric Charge and Electric Field
2 Static Electricity and Charge: Conservation of Charge
Learning objectives.
- Define electric charge, and describe how the two types of charge interact.
- Describe three common situations that generate static electricity.
- State the law of conservation of charge.

What makes plastic wrap cling? Static electricity. Not only are applications of static electricity common these days, its existence has been known since ancient times. The first record of its effects dates to ancient Greeks who noted more than 500 years B.C. that polishing amber temporarily enabled it to attract bits of straw (see (Figure 2.1)). The very word electric derives from the Greek word for amber ( electron ).
Many of the characteristics of static electricity can be explored by rubbing things together. Rubbing creates the spark you get from walking across a wool carpet, for example. Static cling generated in a clothes dryer and the attraction of straw to recently polished amber also result from rubbing. Similarly, lightning results from air movements under certain weather conditions. You can also rub a balloon on your hair, and the static electricity created can then make the balloon cling to a wall. We also have to be cautious of static electricity, especially in dry climates. When we pump gasoline, we are warned to discharge ourselves (after sliding across the seat) on a metal surface before grabbing the gas nozzle. Attendants in hospital operating rooms must wear booties with aluminum foil on the bottoms to avoid creating sparks which may ignite the oxygen being used.
Some of the most basic characteristics of static electricity include:
- The effects of static electricity are explained by a physical quantity not previously introduced, called electric charge.
- There are only two types of charge, one called positive and the other called negative.
- Like charges repel, whereas unlike charges attract.
- The force between charges decreases with distance.
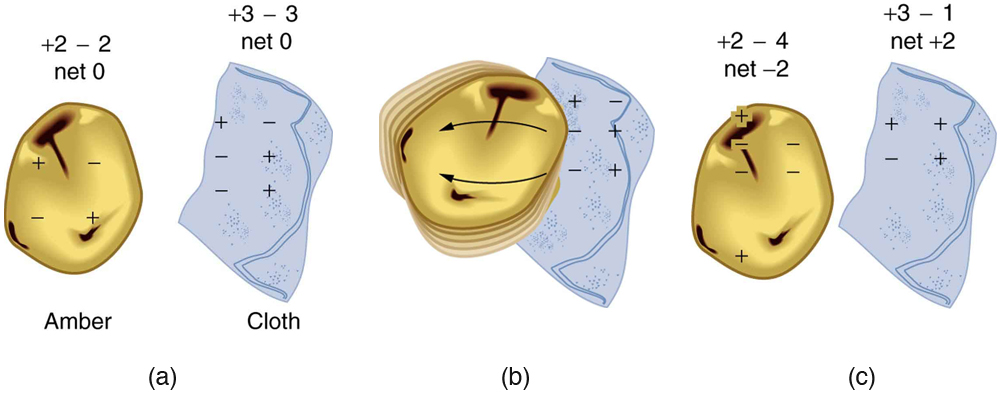
How do we know there are two types of electric charge ? When various materials are rubbed together in controlled ways, certain combinations of materials always produce one type of charge on one material and the opposite type on the other. By convention, we call one type of charge “positive”, and the other type “negative.” For example, when glass is rubbed with silk, the glass becomes positively charged and the silk negatively charged. Since the glass and silk have opposite charges, they attract one another like clothes that have rubbed together in a dryer. Two glass rods rubbed with silk in this manner will repel one another, since each rod has positive charge on it. Similarly, two silk cloths so rubbed will repel, since both cloths have negative charge. (Figure 2.2) shows how these simple materials can be used to explore the nature of the force between charges.
More sophisticated questions arise. Where do these charges come from? Can you create or destroy charge? Is there a smallest unit of charge? Exactly how does the force depend on the amount of charge and the distance between charges? Such questions obviously occurred to Benjamin Franklin and other early researchers, and they interest us even today.
Charge Carried by Electrons and Protons
Franklin wrote in his letters and books that he could see the effects of electric charge but did not understand what caused the phenomenon. Today we have the advantage of knowing that normal matter is made of atoms, and that atoms contain positive and negative charges, usually in equal amounts.
(Figure) shows a simple model of an atom with negative electrons orbiting its positive nucleus. The nucleus is positive due to the presence of positively charged protons . Nearly all charge in nature is due to electrons and protons, which are two of the three building blocks of most matter. (The third is the neutron, which is neutral, carrying no charge.) Other charge-carrying particles are observed in cosmic rays and nuclear decay, and are created in particle accelerators. All but the electron and proton survive only a short time and are quite rare by comparison.
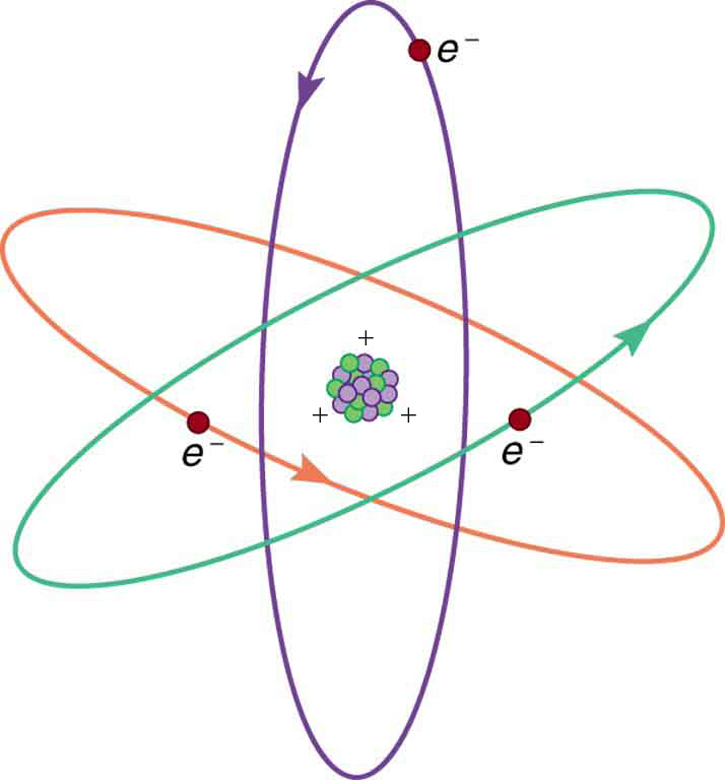
The charges of electrons and protons are identical in magnitude but opposite in sign. Furthermore, all charged objects in nature are integral multiples of this basic quantity of charge, meaning that all charges are made of combinations of a basic unit of charge. Usually, charges are formed by combinations of electrons and protons. The magnitude of this basic charge is
The symbol [latex]q[/latex] is commonly used for charge and the subscript [latex]e[/latex] indicates the charge of a single electron (or proton).
The SI unit of charge is the coulomb (C). The number of protons needed to make a charge of 1.00 C is
Similarly, [latex]6\text{.}\text{25}×{\text{10}}^{\text{18}}[/latex] electrons have a combined charge of −1.00 coulomb. Just as there is a smallest bit of an element (an atom), there is a smallest bit of charge. There is no directly observed charge smaller than [latex]\mid {q}_{e}\mid[/latex] (see Things Great and Small: The Submicroscopic Origin of Charge ), and all observed charges are integral multiples of [latex]\mid {q}_{e}\mid[/latex].
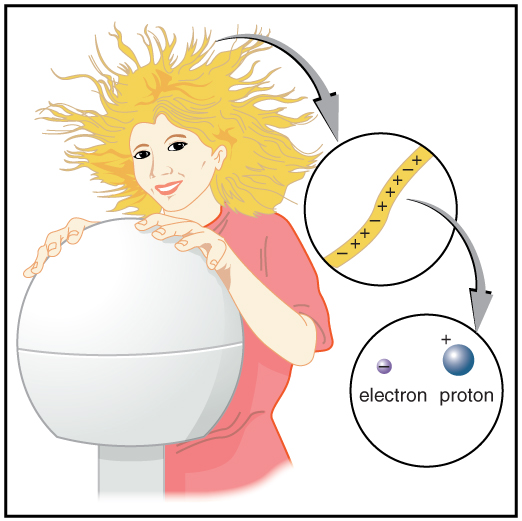
With the exception of exotic, short-lived particles, all charge in nature is carried by electrons and protons. Electrons carry the charge we have named negative. Protons carry an equal-magnitude charge that we call positive. (See (Figure 2.4).) Electron and proton charges are considered fundamental building blocks, since all other charges are integral multiples of those carried by electrons and protons. Electrons and protons are also two of the three fundamental building blocks of ordinary matter. The neutron is the third and has zero total charge.
(Figure 2.4) shows a person touching a Van de Graaff generator and receiving excess positive charge. The expanded view of a hair shows the existence of both types of charges but an excess of positive. The repulsion of these positive like charges causes the strands of hair to repel other strands of hair and to stand up. The further blowup shows an artist’s conception of an electron and a proton perhaps found in an atom in a strand of hair.
The electron seems to have no substructure; in contrast, when the substructure of protons is explored by scattering extremely energetic electrons from them, it appears that there are point-like particles inside the proton. These sub-particles, named quarks, have never been directly observed, but they are believed to carry fractional charges as seen in (Figure 2.5). Charges on electrons and protons and all other directly observable particles are unitary, but these quark substructures carry charges of either [latex]-\frac{1}{3}[/latex] or [latex]+\frac{2}{3}[/latex]. There are continuing attempts to observe fractional charge directly and to learn of the properties of quarks, which are perhaps the ultimate substructure of matter.
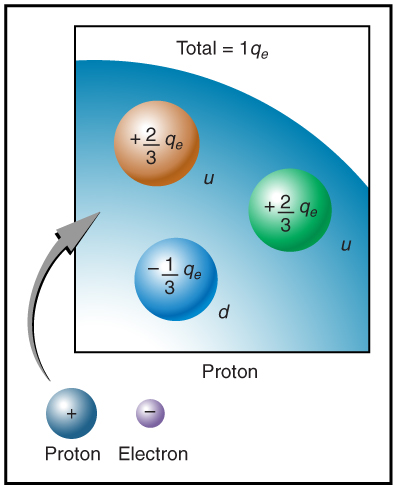
Separation of Charge in Atoms
Charges in atoms and molecules can be separated—for example, by rubbing materials together. Some atoms and molecules have a greater affinity for electrons than others and will become negatively charged by close contact in rubbing, leaving the other material positively charged. (See (Figure 2.6).) Positive charge can similarly be induced by rubbing. Methods other than rubbing can also separate charges. Batteries, for example, use combinations of substances that interact in such a way as to separate charges. Chemical interactions may transfer negative charge from one substance to the other, making one battery terminal negative and leaving the first one positive.
No charge is actually created or destroyed when charges are separated as we have been discussing. Rather, existing charges are moved about. In fact, in all situations the total amount of charge is always constant. This universally obeyed law of nature is called the law of conservation of charge .
Total charge is constant in any process.
In more exotic situations, such as in particle accelerators, mass, [latex]\Delta m[/latex], can be created from energy in the amount
[latex]\Delta m=\frac{E}{{c}^{2}}[/latex]. Sometimes, the created mass is charged, such as when an electron is created. Whenever a charged particle is created, another having an opposite charge is always created along with it, so that the total charge created is zero. Usually, the two particles are “matter-antimatter” counterparts. For example, an antielectron would usually be created at the same time as an electron. The antielectron has a positive charge (it is called a positron), and so the total charge created is zero. (See (Figure 2.7).) All particles have antimatter counterparts with opposite signs. When matter and antimatter counterparts are brought together, they completely annihilate one another. By annihilate, we mean that the mass of the two particles is converted to energy E , again obeying the relationship [latex]\Delta m=\frac{E}{{c}^{2}}[/latex]. Since the two particles have equal and opposite charge, the total charge is zero before and after the annihilation; thus, total charge is conserved.
Only a limited number of physical quantities are universally conserved. Charge is one—energy, momentum, and angular momentum are others. Because they are conserved, these physical quantities are used to explain more phenomena and form more connections than other, less basic quantities. We find that conserved quantities give us great insight into the rules followed by nature and hints to the organization of nature. Discoveries of conservation laws have led to further discoveries, such as the weak nuclear force and the quark substructure of protons and other particles.
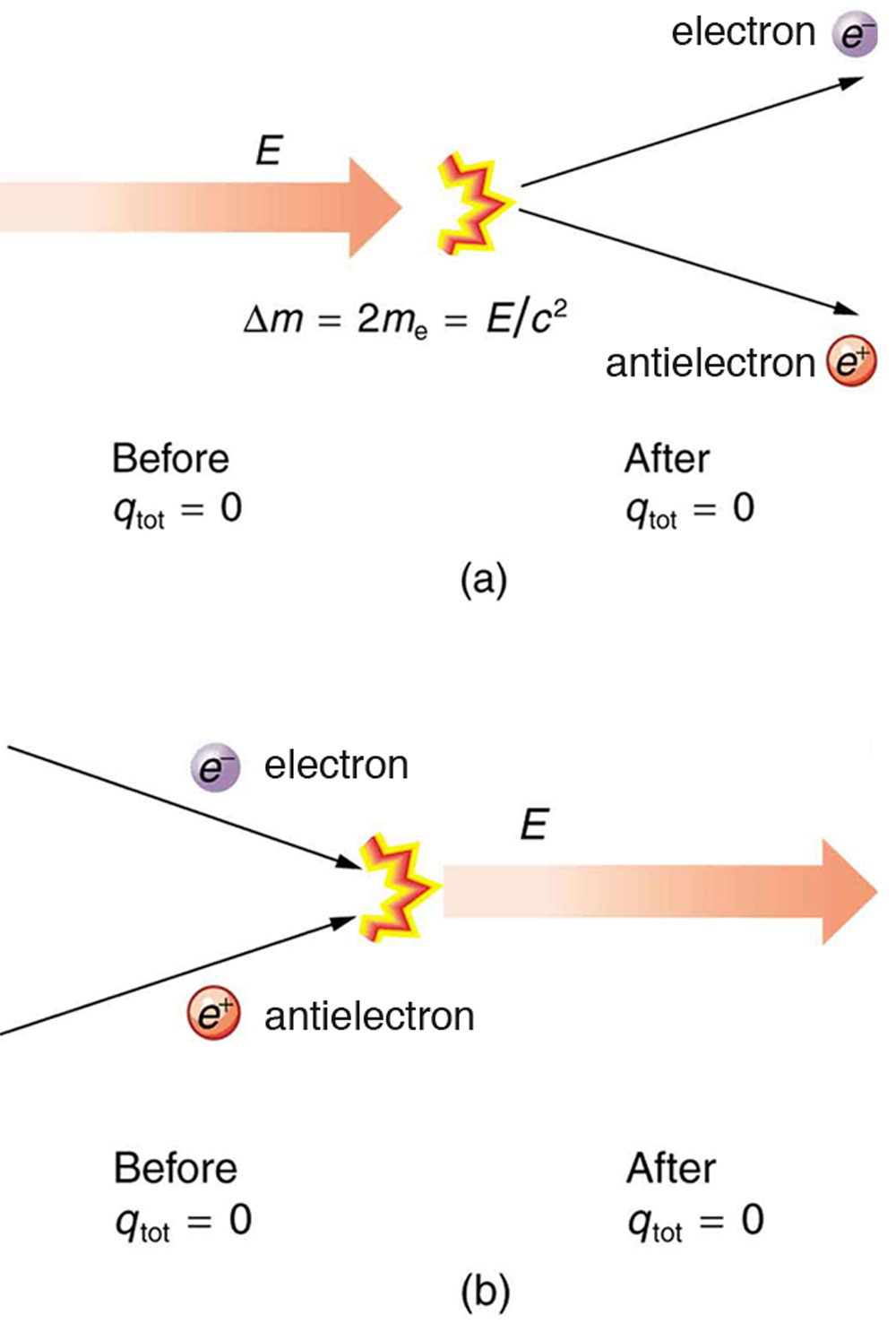
The law of conservation of charge is absolute—it has never been observed to be violated. Charge, then, is a special physical quantity, joining a very short list of other quantities in nature that are always conserved. Other conserved quantities include energy, momentum, and angular momentum.
Why does a balloon stick to your sweater? Rub a balloon on a sweater, then let go of the balloon and it flies over and sticks to the sweater. View the charges in the sweater, balloons, and the wall.
Section Summary
- There are only two types of charge, which we call positive and negative.
- Like charges repel, unlike charges attract, and the force between charges decreases with the square of the distance.
- The vast majority of positive charge in nature is carried by protons, while the vast majority of negative charge is carried by electrons.
- The electric charge of one electron is equal in magnitude and opposite in sign to the charge of one proton.
- An ion is an atom or molecule that has nonzero total charge due to having unequal numbers of electrons and protons.
- The SI unit for charge is the coulomb (C), with protons and electrons having charges of opposite sign but equal magnitude; the magnitude of this basic charge [latex]\mid {q}_{e}\mid[/latex] is [latex]\mid {q}_{e}\mid =1.60×{\text{10}}^{-\text{19}}\phantom{\rule{0.25em}{0ex}}\text{C}.[/latex]
- Whenever charge is created or destroyed, equal amounts of positive and negative are involved.
- Most often, existing charges are separated from neutral objects to obtain some net charge.
- Both positive and negative charges exist in neutral objects and can be separated by rubbing one object with another. For macroscopic objects, negatively charged means an excess of electrons and positively charged means a depletion of electrons.
- The law of conservation of charge ensures that whenever a charge is created, an equal charge of the opposite sign is created at the same time.
Conceptual Questions
There are very large numbers of charged particles in most objects. Why, then, don’t most objects exhibit static electricity?
Why do most objects tend to contain nearly equal numbers of positive and negative charges?
Problems & Exercises
Common static electricity involves charges ranging from nanocoulombs to microcoulombs. (a) How many electrons are needed to form a charge of [latex]–2.00\phantom{\rule{0.25em}{0ex}}\text{nC}[/latex] (b) How many electrons must be removed from a neutral object to leave a net charge of [latex]0.500\phantom{\rule{0.25em}{0ex}}µ\text{C}[/latex]?
(a) [latex]1.25×{\text{10}}^{\text{10}}[/latex]
(b) [latex]3.13×{\text{10}}^{\text{12}}[/latex]
If [latex]1\text{.}\text{80}×{\text{10}}^{\text{20}}[/latex] electrons move through a pocket calculator during a full day’s operation, how many coulombs of charge moved through it?
To start a car engine, the car battery moves [latex]3\text{.}\text{75}×{\text{10}}^{\text{21}}[/latex] electrons through the starter motor. How many coulombs of charge were moved?
A certain lightning bolt moves 40.0 C of charge. How many fundamental units of charge [latex]\mid {q}_{e}\mid[/latex] is this?
College Physics 2 Copyright © 2012 by OSCRiceUniversity is licensed under a Creative Commons Attribution 4.0 International License , except where otherwise noted.
Share This Book
FREE K-12 standards-aligned STEM
curriculum for educators everywhere!
Find more at TeachEngineering.org .
- TeachEngineering
- Take Charge! All About Static Electricity
Lesson Take Charge! All About Static Electricity
Grade Level: 4 (3-5)
Time Required: 45 minutes
Lesson Dependency: Lights Out!
Subject Areas: Physical Science
NGSS Performance Expectations:

- Print lesson and its associated curriculum
Curriculum in this Unit Units serve as guides to a particular content or subject area. Nested under units are lessons (in purple) and hands-on activities (in blue). Note that not all lessons and activities will exist under a unit, and instead may exist as "standalone" curriculum.
- Static Cling
- Charge It! All About Electrical Attraction and Repulsion
- Build a Charge Detector
- Completing the Circuit
- Two-Cell Battery
- Will It Conduct?
- Materials Switcheroo: Construct Simple Electrical Switches
- Bulbs & Batteries in a Row
- Light Your Way: Design-Build a Series Circuit Flashlight
- Build a Toy Workshop
- Bulbs & Batteries Side by Side
| Unit | Lesson | Activity |
TE Newsletter
Engineering connection, learning objectives, worksheets and attachments, more curriculum like this, pre-req knowledge, introduction/motivation, associated activities, lesson closure, vocabulary/definitions, user comments & tips.

Engineers are so creative: Would you ever think that the air we breathe could be cleaned by using the behavior of electrical charges? Engineers who fully understand the scientific principles of static electricity have invented many modern products and industrial processes that help our environment. For example, different types of electrostatic precipitators clean both factory and home air, and electronic paper can be used for communication and re-used over and over again.
After this lesson, students should be able to:
- Understand the basic properties of electric charge.
- Describe technologies associated with static electricity.
- Explain the transfer of electrons between two objects.
- Compare different objects based on their electric charge.
Educational Standards Each TeachEngineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards. All 100,000+ K-12 STEM standards covered in TeachEngineering are collected, maintained and packaged by the Achievement Standards Network (ASN) , a project of D2L (www.achievementstandards.org). In the ASN, standards are hierarchically structured: first by source; e.g. , by state; within source by type; e.g. , science or mathematics; within type by subtype, then by grade, etc .
Ngss: next generation science standards - science.
| NGSS Performance Expectation | ||
|---|---|---|
| 3-PS2-3. Ask questions to determine cause and effect relationships of electric or magnetic interactions between two objects not in contact with each other. (Grade 3) Do you agree with this alignment? Thanks for your feedback! | ||
| This lesson focuses on the following aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Ask questions that can be investigated based on patterns such as cause and effect relationships. Alignment agreement: Thanks for your feedback! | Electric, and magnetic forces between a pair of objects do not require that the objects be in contact. The sizes of the forces in each situation depend on the properties of the objects and their distances apart and, for forces between two magnets, on their orientation relative to each other. Alignment agreement: Thanks for your feedback! | Cause and effect relationships are routinely identified, tested, and used to explain change. Alignment agreement: Thanks for your feedback! |
| NGSS Performance Expectation | ||
|---|---|---|
| 4-PS3-2. Make observations to provide evidence that energy can be transferred from place to place by sound, light, heat, and electric currents. (Grade 4) Do you agree with this alignment? Thanks for your feedback! | ||
| This lesson focuses on the following aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Make observations to produce data to serve as the basis for evidence for an explanation of a phenomenon or test a design solution. Alignment agreement: Thanks for your feedback! | Energy can be moved from place to place by moving objects or through sound, light, or electric currents. Alignment agreement: Thanks for your feedback! Energy is present whenever there are moving objects, sound, light, or heat. When objects collide, energy can be transferred from one object to another, thereby changing their motion. In such collisions, some energy is typically also transferred to the surrounding air; as a result, the air gets heated and sound is produced.Alignment agreement: Thanks for your feedback! Light also transfers energy from place to place.Alignment agreement: Thanks for your feedback! Energy can also be transferred from place to place by electric currents, which can then be used locally to produce motion, sound, heat, or light. The currents may have been produced to begin with by transforming the energy of motion into electrical energy.Alignment agreement: Thanks for your feedback! | Energy can be transferred in various ways and between objects. Alignment agreement: Thanks for your feedback! |
International Technology and Engineering Educators Association - Technology
View aligned curriculum
Do you agree with this alignment? Thanks for your feedback!
State Standards
Colorado - science.
atoms, electrons, electric charge
Have a student volunteer rub a balloon on a sweater or piece of wool and then hold the balloon close to their hair. Observe that the student's hair stands on end.
Challenge the rest of the students in the class to explain why this happens using the information from the TeachEngineering Lesson Lights Out! regarding the movement of electrons and charged objects. Ask the students: Why do you think that your hair stands up after rubbing it with a balloon? (Answer: Electrons move from your hair to the balloon, causing each of the hairs to have the same positive charge. Since objects with the same charge repel each other, the hairs try to get as far from each other as possible. The farthest they can get is by standing up and away from the others.)
Next, ask the students for examples from their own experiences of the buildup or discharge of static electricity. (Possible answers: Walking across the carpet [buildup] and touching a door knob [discharge].) Students can conduct their own experiment with the associated activity Charge It! All About Electrical Attraction and Repulsion .
Lesson Background and Concepts for Teachers
Electric Charge Basics
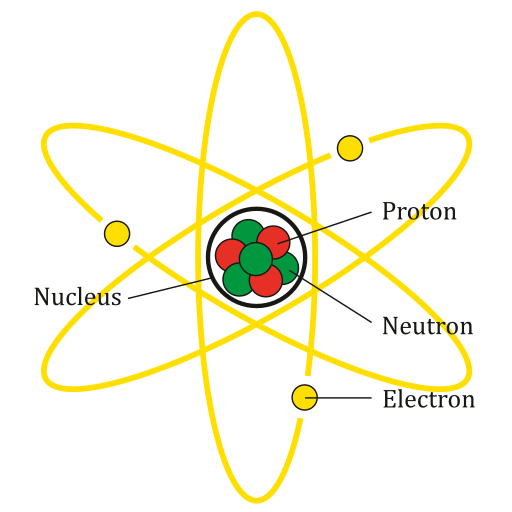
Every atom is made of negatively-charged electrons surrounding a positively-charged nucleus. The nucleus contains protons, which are positively charged, and neutrons, which are neutral (they have no net electric charge), as illustrated in Figure 3. Electrons can move from one atom, molecule or material, to another. Most objects do not have an electric charge because there is a balance of electrons and protons in the material that makes up the object. In certain circumstances, there can be an imbalance of protons and electrons. An object with a greater number of electrons than protons is negatively charged. An object with more protons than electrons is positively charged.
Conductors and Insulators
Materials can be classified by how well they conduct electricity. Insulators do not conduct very well, while conductors do. In insulators, every electron is attached to a particular atom. However, in good conductors, some electrons are more loosely bound to the atom and can move about in the material. Metals are good conductors, while materials such as wood, glass, plastics and rubber are good insulators. Electrical and mechanical engineers use their understanding of the differences in material properties when they design the safe appliances and machines we use every day, such as toasters, coffee makers, hair dryers, stoves and extension cords. Conduct the fun and hands-on associated activity Build a Charge Detector to develop students' understanding of different methods of charging, and the differences between conductors and insulators.
Charging by Friction
When two materials are rubbed together, some electrons may be transferred from one material to the other, leaving them both with a net electric charge. The material that lost electrons becomes positively charged, while the material that gained electrons becomes negatively charged. Both insulators and conductors can gain a net charge in this way. This is how clothing gets charged in the clothes dryer, or our bodies get charged when we walk across a carpeted floor.
Charging by Contact
We can also charge a neutral object (either a conductor or insulator) by just touching it to a charged object. Electrons move from a negatively-charged object to a neutral object, leaving the neutral object with a negative charge. Electrons also move from a neutral object to a positively-charged object, such that the originally neutral object gains a net positive charge. The final charge on the originally-charged object depends on a number of factors, and is not important for what we are studying today. Charging by close proximity is another way to give a neutral conductor a net charge. However, it requires two steps and will not be covered in this lesson.
Excess charge on an object may slowly leak off the object (especially if it is very humid), or the excess charge may produce an electric spark. If a spark is created, electrons move off one object and onto another. During the spark, energy is also released in the form of light, heat and sound. The buildup of excess charge on an insulated body and the resulting spark (or electric discharge) caused by this buildup, are both properties of static electricity.
NOTE: Since in this lesson we explore the attractive force between unlike charges and the repulsion between like charges, some students might have questions about the apparent contradiction of the arrangement of the atom. Students may ask why the positively-charged protons do not repel each other and fly out of the nucleus. The protons in the nucleus are held together in spite of their electrical repulsion because of a stronger nuclear force that works at the atomic scale. Students may also wonder how the electrons keep from spinning into the nucleus. The reasons for this are more complicated; it would probably be best to say that it is similar to why the moon is not pulled into the Earth; in fact, the moon is held in orbit by the Earth.
Electronic Paper
Engineers used the principles of static electricity to develop electronic paper . Someday, paper may be obsolete due to the use of electronic paper technology. Newspapers, bestsellers and textbooks may be printed on electronic paper. This technology could greatly reduce paper production and consequently reduce the forest destruction that results from manufacturing large quantities of paper that our society uses. Electronic paper technology is based on the attractive and repulsive forces between charged objects. Engineers developed electronic paper technology and designed the manufacturing processes that produce electronic paper. Electronic paper is already being used for billboards and signs in stores. Electronic "paper" is thin and flexible like traditional paper, but can be erased and reused over and over again like a computer display.
How does electronic paper work? There are two types (see Figure 3). In one product, the "ink" consists of tiny beads that are black on one side (positively charged) and white on the other (negatively charged). In another type of electronic paper, the "ink" is made of microcapsules containing blue (negatively charged) and white (positively charged) dye particles. In both products, the surface under the layer of ink has tiny electrodes that can be either positively- or negatively-charged. When a portion of the surface is charged, the ink experiences an electric force. If the electrode is positively charged, the positive side of a bead or the white dye particles in the microcapsule are repelled. At the same time, the negative side or the blue dye particles in the microcapsule are attracted to the electrode. This produces a different effect for each type of ink. The image stays the same until the charge on the electrodes is changed.
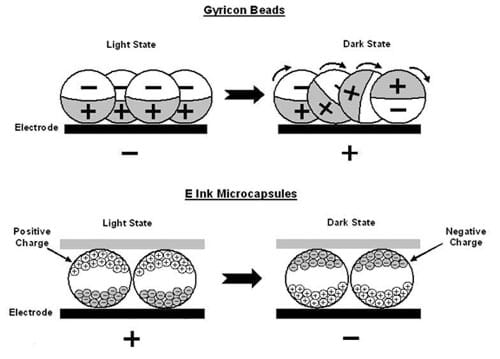
Additional Technologies
Scientists and engineers use their understanding of static electricity to develop industrial air filters to help our environment. These "electrostatic precipitators" use static electricity to remove pollutants without impeding the production efficiency of an industrial plant. Power plants, steel mills and paper plants use electrostatic precipitators to remove the harmful particles generated in the manufacturing process before they can pollute our air.
Electrostatic air cleaners for the home use an electrostatic force to move air molecules and trap small airborne particles (.05 – 30 microns in size, such as pet dander or other allergens) as they circulate past an array of electrically-charged stainless steel blades. One example is the Ionic Breeze by Zenion Effect Technology.
Other technologies that exploit the properties of static electricity may be found in appliances and machines such as copy machines and printers designed by electrical and mechanical engineers.
Watch this activity on YouTube
Ask the students to explain how an object can become electrically charged. Have the students make a list of conductors and insulators on the board. (Possible answers: Insulators could be wood, plastic, Styrofoam. Conductors could be metals.)
atom: The smallest unit of an element, consisting of a dense, positively-charged nucleus surrounded by electrons.
conductor: A material through which charge moves easily.
electrode: A solid conductor through which a current enters or leaves a medium.
electron: The negatively-charged particle of an atom.
Insulator: A material through which charge does not move easily.
ion: An atom, group of atoms or molecule that has a net electric charge.
molecule: The smallest particle of a substance that retains the chemical and physical properties of the substance, and is composed of two or more atoms.
neutron: The neutral particle in an atom.
nucleus: The positively-charged central region of an atom composed of neutrons and protons.
proton: The positively-charged particle of an atom.
static electricity: The buildup of charge on an object, or the spark that is a result of a buildup of charge on an object.
Pre-Lesson Assessment
Discussion Questions: Solicit, integrate and summarize student responses.
- Why do you think that your hair stands up after rubbing it with a balloon? (Answer: Electrons move from your hair to the balloon. Then, each of the hairs has the same positive charge. Since objects with the same charge repel each other, the hairs try to get as far from each other as possible, which is by standing up and away from the other hairs.)
- What are some examples, from your own experiences, of the buildup or discharge of static electricity? (Possible answers: Walking across the carpet [buildup] and touching a door knob [discharge].)
Post-Introduction Assessment
Brainstorming: As a class, have the students engage in open discussion. Remind students that in brainstorming, no idea or suggestion is "silly." All ideas should be respectfully heard. Encourage wild ideas and discourage criticism of ideas. Have them raise their hands to respond. Write ideas on the board. Ask the students:
- Engineers used the principles of static electricity to develop electronic paper. Where could we use electronic paper in this classroom? (Possible ideas: Textbooks, bulletin board, notebook paper, report cards, etc.)
Lesson Summary Assessment
Bingo: Provide each student with a list of the lesson vocabulary terms. Have each student walk around the room and find a student who can define one vocabulary term. Students must find a different student for each word. When a student has all terms completed they shout "Bingo!" Continue until most students have a bingo. Ask the students who shouted "Bingo!" to give definitions of the vocabulary terms.
Homework Worksheet: Assess students' understanding of the lesson by assigning the Take Charge! Homework Worksheet as a take-home quiz. See answers in the Take Charge! Homework Worksheet Answers .

Lesson Extension Activities
Electronic Paper Message Activity : Have students explore electronic paper technology by conducting an activity in which they spell out words using the plus and minus charges on a graph paper grid.
Explore the Van de Graaf generator. Show the students the photograph at the beginning of this lesson, discuss why the person's hair is standing on end. The young person is touching a Van de Graaf generator, which is used to generate static electricity. Developed by an MIT professor, Dr. Robert J. Van de Graaf, they were originally used as research tools in atom-smashing experiments, but now are mainly used to teach people about static electricity and lightning. Note that the person must be standing on an insulator to prevent charge from moving through her body and going into the Earth. Also, they must be very careful not to touch anyone because that would give them a very unpleasant electric shock. Ask students to research how the Van de Graaf generator works. What type of charging occurs in a Van de Graaf generator?
Have students research the history of photocopying. American law student Chester Carlson was a research engineer and a patent attorney who invented the method of xerography — a copy process based on electrostatic energy. Students can create a timeline of the changes in photocopying technology since Chester's time.
Introduce the students to accomplished science researchers such as Marietta Blau (1894-1970). She was an Austrian physicist and pioneer in the study of atomic particles, and the first person to develop methods for identifying both protons and neutrons in experiments, using photographic chemicals. Because she was Jewish, Marietta Blau fled from Austria before the Anschluss. In the U.S., she continued her research in industry and at universities, including Brookhaven National Laboratories, Columbia University and the University of Miami. She was also a professor at the Technical University of Mexico City. Several Nobel Prize-winning scientists nominated Marietta Blau for the Nobel Prize in Physics.

This lesson introduces the concept of electricity by asking students to imagine what their life would be like without electricity. Students learn that electrons can move between atoms, leaving atoms in a charged state.

Students explore the static electricity through hands-on activities. They attract O-shaped cereal pieces to charged combs and watch the cereal jump away when it touches the comb. They also observe Styrofoam pellets pulling towards a charged comb, then leaping back to the table.

Students are introduced to the concept of electricity by identifying it as an unseen, but pervasive and important presence in their lives. They compare conductors and insulators based on their capabilities for electron flow. Then water and electrical systems are compared as an analogy to electrical ...

Students are introduced to the fundamental concepts of electricity. They address questions such as "How is electricity generated?" and "How is it used in every-day life?" Illustrative examples of circuit diagrams are used to help explain how electricity flows.

Ditlea, Steve. "The Electronic Paper Chase." Scientific American. November 2001 issue.
Electrical Engineering for Pollution Control, Electrostatic Precipitator for Power Plants, ASU Electrical Engineering.
Electronic ink technology, E Ink Corporation.
Galison, Peter L. "Marietta Blau: Between Nazis and Nuclei." Physics Today. Vol. 50. Issue 42 1997.
Hewitt, Paul G. Conceptual Physics. 8th Edition. New York, NY: Addison Publishing Company, 1998.
Patsuris, Penelope. "Marketing Messages Made to Order." Forbes, August 27, 2003.
Contributors
Supporting program, acknowledgements.
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: January 28, 2021
Static Electricity Revision Notes, Past Papers & Questions for Leaving Cert Physics
Join thousands of students using SimpleStudy to revise for their for Leaving Cert.

48547 happy Leaving Cert students

Study Physics Past Papers & Marking Schemes
SimpleStudy’s Leaving Cert Physics Past Papers cover all of Static Electricity. Revise past papers and marking schemes, or just Static Electricity questions.
Static Electricity Revision Notes
Study beautiful, concise and carefully crafted notes, for your Leaving Cert.
Static Electricity Questions
Past Questions, timed, divided by topic and automatically graded.
Static Electricity Quizzes & Flashcards
Make studying easy with multiple choice quizzes and fun flashcards.
Practice Revision Questions For Static Electricity
Test your knowledge! Try SimpleStudy’s interactive revision questions, made specifically for every topic and note so you’re properly prepared for your Leaving Cert Physics exam.
83% of students got this correct.
What happens when two charged rods are brought close to each other?
Leaving cert physics static electricity revision notes.
Static Electricity revision notes, study carefully curated revision materials, broken down into focused topics.

Learn how to study Leaving Cert Physics .
Leaving Cert Static Electricity questions and answers.
Reviewed by subject experts and top students.
Example Static Electricity answers
Example answers, on past Static Electricity Questions. Learn from other students with SimpleStudy’s example answers and essays.

Graded, marked example answers for every question.
Sample essays from A+ students.
Experiment write-ups, practicals, diagrams and more.

Scan your Static Electricity Homework to get instant feedback and grading.
Upload a photo of your Static Electricity homework or question and get feedback instantly using our AI.
Exam-focused quizzes for Static Electricity
Fun and easy Static Electricity quizzes based on Leaving Cert Physics past papers. Practice multiple choice questions, see explanations for every answers, and track your progress.

Over 2 quiz questions on Static Electricity.
94% of students improved their grades.
The best way to revise for class tests.

Try our Static Electricity flashcards, or create your own.
Join the leaving cert physics students..., 97% of students.
Report Improved Results
98% of Students
Recommend to friends
Students Supported
Questions answered
Students love Leaving Cert Physics Revision
SimpleStudy makes revision simple, with easy to study resources and PDF’s for every level and every topic.
Google Reviews and Trustpilot

Other Leaving Cert Physics topics to explore
Circular motion, electrical circuits, electromagnetic induction, heat & heat transfer, nuclear energy, particle physics, potential difference & capacitance, reflection & mirrors, refraction & lenses, semi-conductors, simple harmonic motion ( shm ), speed & velocity, temperature, the atom & radioactivity, the electron, vibration & sound, wave motion, work, energy & power, acceleration, current & charge.

We think you are located in Russian Federation . Is this correct?
- Yes, I reside in Russian Federation
- Change country/curriculum
We use this information to present the correct curriculum and to personalise content to better meet the needs of our users.
9.1 Friction and static electricity
Static electricity
Chapter overview
In previous grades the learners investigated circuits and current electricity. In this chapter they are introduced to static electricity. It explains how static electricity is caused by friction between objects and that charged objects are either positively or negatively charged. There are several activities in this chapter which illustrate the effects of static electricity.
An interesting article on how to encourage learners to pursue STEM (Science, Technology, Engineering and Mathematics) careers: [link] http://spectrum.ieee.org/at-work/education/the-stem-crisis-is-a-myth
1.1 Friction and static electricity (3 hours)
|
|
|
|
| Activity: Sticky balloons | Observing, working in pairs | Suggested |
| Activity: Turning the wheel | Observing, recording | CAPS suggested |
| Activity: Research the practical applications of static electricity | Researching, writing, summarising | Optional |
| Activity: Making a simple electroscope | Carrying out instructions, observing, predicting, explaining | Optional |
- What is static electricity?
- What is friction?
- Why does my hair stand on end and crackle when I pull a jersey off?
- What is lightning?
- What does it mean to 'earth' an object?
- What does it mean when we say 'opposites attract'?
Have you ever pushed a trolley through the shops and suddenly felt a shock? Or pulled your school jersey over your head and heard it crackling? What causes those shocks and noises? Let's investigate.
Friction and static electricity
- static electricity
- electrostatic charge
- attract: to pull something closer
- repel: to push something away
The effects of static electricity are all around us, but we do not always recognise it when we see or feel them. Or perhaps you have, but you never realised what was causing it. For example, have you ever felt a slight shock when you put a jersey over your head on a cold day, or perhaps you have observed your hair stand on end when you touch certain objects? Let's do a quick activity to demonstrate static electricity.
Watch this video about static electricty to understand why your hair stands on ends when you brush it or rub it against a balloon
Sticky balloons
You can also do this activity using a plastic comb rather than balloons. Or else you can use pieces of paper instead of a learner's hair as not all hair will behave in the following way if it has product in it. You can then rather rub the balloon on a jersey and pick up pieces of paper.
- balloons (or a plastic comb)
- small pieces of paper
INSTRUCTIONS:
Hold the balloon a short distance away from your hair or pieces of paper. What do you notice?
Nothing happens.
Now hold the balloon a short distance away from your hair or pieces of paper. What do you see?
The hair should "rise" and stick to the balloon, or the pieces of paper will stick to the balloon.

Rubbed it vigorously with the balloon.
Let's look at an everyday example of static electricity. Sometimes when you comb your hair with a plastic comb your hair stands on end and makes crackling sounds. How does this happen?
You have dragged the surface of the plastic comb against the surfaces of your hair. When two surfaces are rubbed together there is friction between them. Friction is a resistance against the movement of an object as a result of its contact with another object. This means that when you rubbed the plastic comb along your hair, your hair resisted the movement of the comb and slowed it down.
The friction between two surfaces can cause electrons to be transferred from one surface to the other.
In order to understand how electrons can be transferred, we need to remember what we learned about the structure of an atom last term in Matter and Materials.

All atoms have a nucleus which contains protons and neutrons. The nucleus is held together by a very strong force, which means that the protons within a nucleus can be considered to be fixed there. The atom also contains electrons. Where are the electrons arranged in the atom?
The electrons are arranged in the space around the nucleus.
What is the charge on a proton?
Positive charge.
What is the charge on an electron?
Negative charge.
What is the charge on a neutron?
Neutrons are not charged. They are neutral.
The atom is held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electrons. Within an atom, the electrons closest to the nucleus are the most strongly held, whilst those further away experience a weaker attraction.
Normally, atoms contain the same number of protons and electrons. This means that atoms are normally neutral because they have the same number of positive charges as negative charges, so the charges balance each other out. All objects are made up of atoms and since atoms are normally neutral, objects are also usually neutral.
However, when we rub two surfaces together, like when you comb your hair or rub a balloon against your hair, the friction can cause electrons to be transferred from one object to another. Remember, the protons are fixed in place in the nucleus and so they cannot be transferred between atoms, it is only electrons that are able to be transferred to another surface. Some objects give up electrons more easily than other objects. Look at the following diagram which explains how this happens.

Which object gave up some of its electrons in the diagram?
Does this object now have more positive or more negative charges?
It has more positive charges.
Which object gained electrons in the diagram?
It has more negative charges.
When an object has more electrons than protons overall, then we say that the object is negatively charged .
When an object has fewer electrons than protons overall, then we say that the object is positively charged .
Have a look at the following diagram which illustrates this.

So, we now understand the transfer of electrons that takes place as a result of friction between objects. But, how did that result in your hair rising when you brought the charged balloon close to your hair in the last activity? Let's look at what happens when oppositely charged objects are brought together.
Turning the wheel
This is a fun demonstration of how like charges repel each other and unlike charges attract each other. If you have enough materials, allow the learners to try this themselves. If you don't have enough materials, do this as a demonstration but give the learners a chance to play a bit.
Practise this activity a few times first to make sure that you have the method right. Remember that it is quite easy to accidently earth the rods so work with care. This will work best on a dry day. This will be dependent on the area which you live in.
At a brainstorming workshop with volunteer teachers and academics at the beginning of 2013, we filmed a quick demonstration of this task when the group was discussing it. You can view this short clip here:
- 2 curved watch glasses
- 2 perspex rods
- cloth: wool or nylon
- plastic rod
- small pieces of torn paper
Bring the second perspex rod close to the first perspex rod. What do you see happening?

The second perspex rod should repel the first one as they have like charges, so learners should see the second rod 'pushing' the first one around in a circle.
You might need to rub the first perspex rod again, in between attempts, as the charge does dissipate.
The rods now have opposite charges and so the second rod should be seen to 'pull' the other rod around in a circle.
The learners should be able to pick up the pieces of paper with the charged rod.
What happened when you brought the second perspex rod close to the first perspex rod?
When the rods are the same (i.e. both perspex) then the first rod should move away from the second and the top watch glass will turn in a circle.
What happened when you brought the plastic rod close to the first perspex rod?
When the two different materials are used then the first rod should move towards the plastic rod and the watch glass will turn in a circle towards the plastic rod.
What happened when you brought the plastic rod close to the pieces of paper?
The pieces of paper were attracted to the plastic rod.
When we rubbed the perspex rods with the cloth, electrons were transferred from the perspex to the cloth. What charge do the perspex rods now have?
A positive charge.
Both the perspex rods now have the same charge. Did you notice that objects with the same charge tend to push each other away? We say that they are repelling each other.
When we rubbed the plastic rod with the cloth, electrons were transferred from the cloth to the plastic rod. What charge does the plastic rod now have?
A negative charge.
The perspex rod and the plastic rod now have opposite charges. Did you notice that objects with different charge tend to pull each other together? We say that they are attracting each other.
In the example of the pieces of paper being attracted to the ruler, the paper starts off neutral. However, as the negatively charged plastic rod is brought closer, the electrons in the paper that are nearest to the rod will begin to move away, leaving behind a positive charge on the surfaces of the paper that are nearest to the rod. The paper is therefore attracted to the rod because opposite charges attract. Another example is dust that is attracted to newly polished glasses.
Discover more with a simulation on rubbing balloons and a jersey. http://phet.colorado.edu/en/simulation/balloons
We have now observed the fundamental behaviour of charges.
In summary, we can say:
- If two negatively charged objects are brought close together, then they will repel each other.
- If two positively charged objects are brought close together, then they will repel each other.
- If a positively charged object is brought near to a negatively charged object, they will attract each other.
Remember, like charges repel and opposite charges attract.
Do you now understand why your hair rises and is attracted to the balloon after you rub the balloon on your hair? Write a short description to explain what is happening using the words: electrons, transfer, negative charge, positive charge, opposite, attract, repel.
When rubbing hair with the balloon, electrons are transferred from the hair to the balloon. The balloon now has a negative charge and the hair has a positive charge. They have opposite charges and so when the balloon is brought close to the hair again, they attract each other. Since the hair strands each have positive charges, like charges repel and the hair strands repel each other, also causing them to rise up.
Opposites attract and like repel (video)
Sparks, shocks and earthing
A large build-up of charge on an object can be dangerous. When electrons transfer from a charged object to a neutral object we say that the charged object has discharged.
Discharging can take place when the objects touch each other. But the electrons can also transfer from one object to another when they are brought close, but not touching. When electrons move across an air gap they can heat the air enough to make it glow. The glow is called a spark .

Sparks can be harmless, but they can also be very dangerous. Sparks can cause flammable materials to ignite . You will probably have noticed that you may not smoke cigarettes or have open flames near petrol tanks at petrol stations. This is because petrol fumes are very explosive and only need a small amount of heat to start them burning. A small electrostatic spark is enough to ignite flammable petrol fumes.
A video showing the dangers of sparks of static electricity at a petrol station.
This video in the Visit box shows how static electricity from the flowing petrol causes a spark which ignites the petrol fumes and leads to a large fire. It is an illustration of one of the dangers of static electricity.
Electrostatic discharge can also cause electric shocks . Have you ever been shocked by a shopping trolley while you are pushing it around a shop? Or have you walked across a carpeted room and then shocked yourself when you touch the door handle to leave the room? You have experienced an electric discharge. Electrons move from the door handle onto your skin and the movement of the electrons causes a small electric shock. Small electric shocks can be uncomfortable but mostly harmless. Large electric shocks are extremely dangerous and can cause injury and death.
A simulation on friction between a carpet and John Travolta's foot. http://phet.colorado.edu/en/simulation/travoltage
The discharge of electrons from charged objects happens much more easily when the air is dry, which is why you are more likely to experience electrostatic sparks or shocks in dry weather. This is because when the weather is humid, the moisture in the air can collect on the surface of objects, and prevent the build-up of electrical charge. The charge dissipates through the moisture, which is a better conductor than air.
Do you know where else we can see sparks due to static electricity? Look at the photo for a clue!

During a thunderstorm, there is friction in the atmosphere between the particles that make up clouds, causing the build-up of regions of charge. Once the difference in charge between two regions becomes great enough, electrostatic discharge becomes possible. A lightning flash is a massive discharge between charged regions within clouds, or between clouds and the Earth.
Lightning bolts can travel at about 210 000 km/h and get as hot as 30 000 °C.
How to survive a lightning strike.
In order to discharge extra electrons safely from an object we must earth it. Earthing means that we connect the charged object to the ground (the Earth) with an electrical conductor. The extra electrons travel along the conductor and enter the ground without causing any harm. The Earth is so large that the extra charge does not have any overall effect.
For example, think of the metal trolleys in shopping centres. Have you ever noticed that they normally have a metal chain hanging at the bottom which drags along the floor? This is to earth the trolley if it gets a charge so that charge cannot build up on the trolley. This protects the person pushing the trolley from getting a shock.
Research the practical applications of static electricity
- Use the internet or your school or community library to find information about the practical applications of static electricity.
- Research one useful effect of static electricity and one problem caused by static electricity.
- Write a short paragraph explaining your research.
There are many different useful and damaging effects of static electricity. Here are some examples.
- useful: air filters remove smoke particles; spray painting; photocopying
- problems: dust on TV and computer screens; damage to electronic equipment
We are now going to look at two instruments which demonstrate static electricity.
Van de Graaff generator
If you do not have a Van de Graaff generator then you can use some of the videos provided here which show and explain how the generator works. If you do have a generator then allowing the learners to "play" with it will give them a good insight into the effects of static electricity. Allow learners to perform different activities, such as having their hair stand on end.
Let the learners hold onto the dome and then run the generator until their hair stands on end.
Tear up small pieces of paper and place them on the top of the uncharged dome, run the generator and the pieces will become charged and then fly off the generator. This is a good example of the pieces of paper becoming charged and then, because they all have the same charge, repelling each other.
The Van de Graaff generator is a machine which uses friction to generate a large build-up of electric charge on a metal dome.
Should a person touch 20 000 Volts? Visit this link to find out!
The fundamental idea of using friction in a machine to generate a charge dates back to the 17th century, but the generator was only invented by Robert Van de Graaff in 1929 at Princeton University.
The Van de Graaff generator can be used to demonstrate the effects of an electrostatic charge. The big metal dome at the top becomes positively charged when the generator is turned on. When the dome is charged it can be discharged by bringing another insulated metal sphere close to the dome. The electrons will jump to the dome from the metal sphere and cause a spark.
Watch this video so see how a Van de Graaff generator works

You can also touch the dome and your hair will rise. Why do you think this happens?
When you touch the positively charged dome, electrons are transferred from you to the dome to discharge it. This causes you and your hair to become positively charged. The individual hair strands are then positively charged so they repel each other and stand on end.
Electroscope
An electroscope is an early scientific instrument used to identify the presence of a charged object or it can be used to identify the type of charge on a charged object.

The following images show some drawings of different types of electroscopes.

The electroscope is made up of an earthed metal box with glass windows. There is a metal rod hanging down and at the end are two strips of thin gold foil attached to it. A disc or ball is attached to the top of the metal rod, as seen in the illustrations above. When the metal ball or disc at the top is touched with a charged object, or a charged object is brought near to it, the gold foil strips spread apart, indicating that the object has a charge.
Look at the next illustration which shows how this works.

The positively charged rod attracts electrons to the disc from the gold foil strips. The disc at the top becomes negatively charged and the gold foil strips at the bottom become positively charged. Why do the gold foil strips move apart?
They move apart as they now both have a positive charge and positive charges repel.
You can make a simple electroscope with everyday items. Let's try.
Making a simple electroscope
If you cannot find glass jars with lids then it is possible to make lids. Use old plastic tub lids and cut out a circle the same size as the opening of the glass jar. Then use electrical tape (or even masking tape) to hold the plastic lid in place over the jar opening.
The copper does not have to be 14 gauge but the thicker the piece the better it holds it's shape.
Detailed instructions and videos can be found on the internet. Try video in the Visit box for an excellent description of the method.
Make your own electroscope (video)
- glass jar, with lid
- 14 gauge copper wire, about 12 cm in length
- plastic straw or plastic tubing
- 2 small pieces of aluminium foil
- piece of wool cloth
- plastic ruler
- Twist one end of the copper wire into a spiral shape. This will increase its surface area.
- Make a hole in the jar lid and push a small piece of the plastic tubing through the hole.
- Put the other end of the copper wire through the straw so that the spiral end is on the outside of the lid.
- Make a hook out of the pointed end of the copper wire.
- Cut two rectangular strips of aluminium foil.
- Put each piece of aluminium foil onto the hook. Make a small hole in the aluminium foil to allow it to hang from the hook.
- Carefully put the hook end of the copper wire into the glass jar and close the jar.
- Rub the ruler with the wool cloth for a minute.
- Bring the ruler close to the spiral end of the copper wire.
What did you observe when you brought the ruler close to the copper wire?
The two pieces of aluminium foil moved apart.
What happens if you move the ruler away from the copper wire?
The aluminium foil pieces move back together.
Why do the pieces of aluminium foil move apart? When you rubbed the plastic ruler with the wool cloth, the ruler became negatively charged. When the negatively charged ruler is brought close to the copper wire, the electrons on the wire are repelled downwards towards the aluminium foil. The pieces of aluminium foil then have extra electrons on them and they both become negatively charged. Two objects which are negatively charged will repel each other and so the pieces of aluminium foil move away from each other.
This next question is a test of the learners' understanding of the fact that positive charges do not move to cause charging, only electrons can move. But, a positively charged object can move. Learners often get confused with this. Give them a chance to reason out the answer themselves. Allow them to bring a positively charged object close to the electroscope to observe what happens and then try to figure out why the effect is seemingly the same. Rubbing a glass rod with the wool cloth will cause a positive charge to develop on the glass rod.
Write a short paragraph to explain what would happen if you brought a positively charged object close to your electroscope.
When a positively charged object is brought close to the electroscope the negative electrons are attracted towards the positively charged object and move up through the copper wire. This means that the pieces of aluminium have lost some electrons and so have an overall positive charge. Both pieces of aluminium foil are then positively charged. Like charges repel each other and so the pieces of aluminium foil move apart from each other.
- Objects are usually neutral because they have the same number of positive and negative charges.
- Objects can become negatively or positively charged when friction (rubbing) results in the transfer of electrons between objects.
- Protons and neutrons cannot be transferred, only electrons can be transferred by friction.
- If an object has more electrons than protons, then it is negatively charged.
- If an object has fewer electrons than protons, then it is positively charged.
- Like charges repel each other, i.e. negative repels negative; positive repels positive.
- Opposite charges attract each other, i.e. negative attracts positive; positive attracts negative.
- A discharge of the electrons from a charged object can cause sparks or shocks of static electricity, especially when the air is dry.
Concept map
Complete the following concept map to summarise what you have learned in this chapter about charge and static electricity.

Teacher's version

Complete the following sentences. Just write the missing word on the line below.
An object which has a negative charge is said to have _________electrons than protons. [1 mark]
An object which has a negative charge is said to have more electrons than protons.
An object which has a positive charge is said to have _________electrons than protons. [1 mark]
An object which has a positive charge is said to have fewer electrons than protons.
- gained electrons
- gained protons
- lost electrons
- lost protons
- the perspex rod got extra protons from the cloth.
- the perspex rod got extra protons due to friction.
- protons were created as the result of friction.
- the perspex rod lost electrons to the cloth due to friction.
|
|
|
|
3 marks for each of the scenarios, 1 mark is awarded to the drawing and 2 marks to the explanation.
|
|
|
|
| Learners must draw the spheres moving towards each other. | The spheres have opposite charges, which attract, so they move towards each other. | |
| Learners must draw the spheres moving away from each other. | The spheres have the same, positive charge and like charges repel, so they move away from each other. |
|
|
|
|
3 marks for each of the objects, 1 mark is awarded to the calculation and 2 marks to the explanation.
|
|
|
|
| Charge = 4 + (-4) = 0 | It is neutral as there are equal numbers of positive and negative charges. | |
| Charge = 3 + (-6) = -3 | It is negatively charged as there are 3 more negative than positive charges. | |
| Charge = 7 + (-3) = 4 | It is positively charged as there are 4 more positive charges than negative charges. |
The ruler in this photo has been rubbed with a cloth. Describe what is happening in this photo and why. [4 marks]

Rubbing the ruler with a cloth transfers electrons from the cloth to the ruler so the ruler now has an excess of electrons and it is negatively charged. The pieces of paper are neutral. When the negatively charged ruler is brought near to the paper pieces, they are attracted to the ruler as the the electrons move around on the paper because of the large charge on the ruler. Electrons will move away from the ruler leaving a positive charge on the paper near the ruler, so they are attracted.
Sometimes, when you are pushing a trolley, you can get a small shock. Explain why this would happen. [2 mark]
Friction between the floor and the trolley wheels causes a build-up of charge on the trolley. The charge is earthed by your body, causing the shock.
Why does your jersey make a crackling sound when you pull it over your head? [2 mark]
When you pull the jersey over your head the friction causes the jersey and your hair to become charged. The movement of electrons from your hair to the jersey releases energy in the form of light and sound.
Why do trucks transporting petrol drag a short length of metal chain on the road as they drive? [2 mark]
When the truck is driving the movement of the petrol in the tank causes a build-up of charge which could cause a dangerous spark when the fuel is off-loaded. The chain earths the tank. The excess charge on the tank is allowed to dissipate to the road.
What do you think these two girls are touching on the left of the photo? Explain your answer and what is happening to them. [3 marks]

The girls are touching the hollow dome of a Van de Graaff generator. The dome is positively charged so electrons are transferred from their bodies to the dome to discharge it. This causes their bodies and hair to become positively charged. Their hair strands now repel each other as they are all positive (like charges repel) and they rise up.
Total [32 marks]

COMMENTS
Define electricity. Answer: Electricity is defined as the rate of flow of charges in a conductor. 7. How is electric charge classified? Answer: The electric charge is classified as a positive charge and a negative charge. 8. State true or false: Static electricity is the electric field which is developed due to the stationary charges. TRUE. FALSE.
Static electricity | Causes, Examples, Facts, & Description
It could exert either a repulsive or attractive influence upon neutral paper bits. It has an excess of protons compared to the number of electrons. It could be used to charge an electroscope negatively by the process of induction. It lost negative electrons and gained positive electrons during the charging process.
Some of the most basic characteristics of static electricity include: The effects of static electricity are explained by a physical quantity not previously introduced, called electric charge. There are only two types of charge, one called positive and the other called negative. Like charges repel, whereas unlike charges attract.
Static Electricity Essay. Static Electricity Everything is made up of tiny particles called atoms. The atoms are made up of even smaller parts called protons, electrons and neutrons. Protons have a positive charge (+), electrons have a negative charge (-) and neutrons have no charge at all, they are neutral. Atoms normally have the same amount ...
Static Electricity Example Questions. Question 1: Define "Static Electricity". [2 marks] GCSE Physics Foundation Physics Higher AQA. Show Answer. Question 2: When a cloth is rubbed against an acetate rod, electrons from the rod move onto the cloth.
Electricity: Static Electricity • Static electricity : Static electricity is an unbalanced electric charge • This occurs when there is unbalanced number of neutrons and protons, unlike current electricity that occurs when there are conductors involved • The cause of static electricity is when there is an unbalanced molecular construction or non conductive insulators such as plastic ...
Examples of Static Electricity (Edexcel GCSE Physics)
Summary. Students come to make sense of the phenomena of static electricity as they use the science and engineering practices of asking questions and making observations to learn about the nature of electric charge and different methods for charging objects. In a hands-on activity, students induce an electrical charge on various objects, and ...
Paper 1 (Non-fiction Texts and Transactional Writing) Paper 2 (Poetry and Prose Texts and Imaginative Writing) IGCSE Physics Edexcel Topic Questions 2. Electricity Static Electricity.
Exam-focused quizzes for Static Electricity. Fun and easy Static Electricity quizzes based on Leaving Cert Physics past papers. Practice multiple choice questions, see explanations for every answers, and track your progress. Over 2 quiz questions on Static Electricity. 94% of students improved their grades. The best way to revise for class tests.
Pure water DOES NOT conduct electricity well. Only when there are impurities does water conduct electricity well. PTS: 1 REF: K OBJ: 9.3 LOC: Unit C - PLC5 TOP: Unit C - Ch. 09 KI2 4.ANS: D Friction causes a transfer of charge (rubbing from one to another) as does charging by conduction (charge
948 Words4 Pages. Static electricity can be simply defined as the electricity that comprises of isolated unmoving charges (as those produced by friction). ("Static electricity - Definition and More from the Free Merriam-Webster Dictionary," n.d.) Static electricity is the outcome of an imbalance between negative and positive charges in an ...
9.1 Friction and static electricity
View answer. 1d 2 marks. A man pushes a metal trolley along a corridor towards a lift. The trolley has nylon wheels and the floor of the corridor is covered with plastic. The man wears shoes with rubber soles. As he moves the trolley, the man gains an electric charge. Explain how the man gains an electric charge.
Explain. The shock is caused by the discharge of built-up static electricity. Sailors are more at risk when the fuel is being loaded or unloaded. The vapour from the fuel is highly flammable, increasing the risk of fire. The jumper will be positively charged because it loses electrons to the ruler.
Static electricity is usually created when materials are pulled apart or rubbed together, causing positive (+) charges to collect on one material and negative (−) charges on the other surface. Results from static electricity may be sparks, shocks or materials clinging together. Use of static electricity • Photocopy: Copy machines use.
There are multiple choice and essay questions that follow the passage that include different levels of understanding and require students to refer back to the text, use context clues, and understand the main idea of the text with supporting detail. This resource includes:-static electricity reading passage-10 multiple choice questions
Stuck? View answer. 1b 2 marks. Which of the following are examples of static electricity? Tick ( ) two boxes. Hair sticking up after taking off a hat. Electrons flowing through an electrical circuit. Photocopiers sticking power to a document. High-speed electrons emitted during nuclear decay.
200 Words Essay On Electricity. Electricity is a form of energy that is all around us and plays a vital role in our daily lives. It is the force that powers everything from the lights in our homes to the computers we use at school. Science Behind It | Electricity is a flow of tiny particles called electrons. These electrons flow through wires ...